miophthalmology
New Approved Treatments for Geographic Atrophy: Insights
WRITERS Dr Cheryl Au, Dr Long Phan, and Professor Andrew Chang AM

There are now two approved treatments available in Australia for foveal-sparing geographic atrophy (GA) secondary to age-related macular degeneration (AMD), evidenced by progression. This article explores what this means for clinicians and patients.
Geographic atrophy is the late-stage disease manifestation of non-neovascular AMD that progresses to severe central vision loss, leading to loss of ability to read, drive, and recognise faces (Figure 1). Traditionally, the management of GA has focused on visual rehabilitation, such as magnifying glasses and visual aids, optimisation of risk factors such as smoking and diet, as well as referral to low vision support services.
In 2023, two drugs received United States Food and Drug Administration (FDA) approval as treatments for GA. These were pegcetacoplan (Syfovre, Apellis Pharmaceuticals, Inc.) and avacincaptad pegol (Izervay, Astellas Pharma Inc.). Australia is the first country outside of the United States to approve the use of these medications to treat foveal-sparing GA secondary to AMD, evidenced by progression. Pegcetacoplan was the first to receive Therapeutic Goods Administration (TGA) approval in January 2025, followed by avacincaptad pegol in October 2025. Pegcetacoplan has also received a positive Pharmaceutical Benefits Advisory Committee (PBAC) recommendation, which will make this treatment more financially accessible to Australian patients through the Pharmaceutical Benefits Scheme (PBS) when the listing occurs.
It is important to note that both these therapies do not cure or reverse GA, but rather slow down the rate of GA progression.
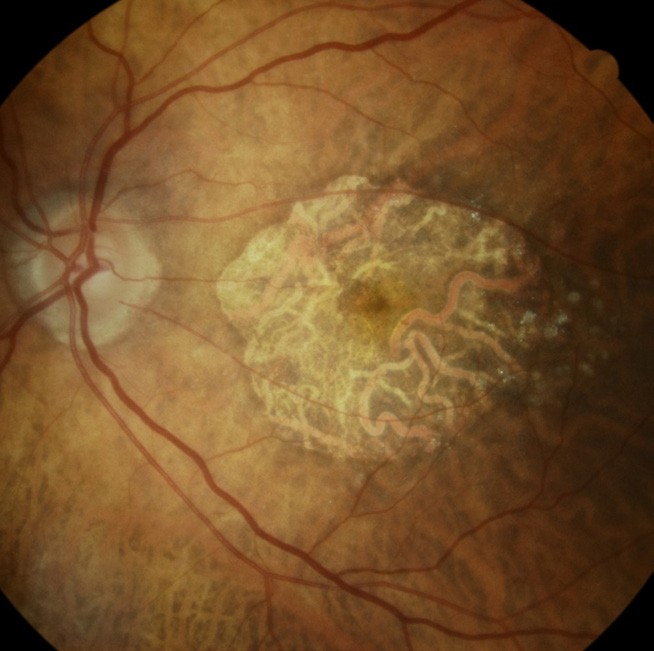
Figure 1. Colour fundus photography of an eye with geographic atrophy.
Both therapies target the complement pathway, which is pathologically overactive in GA. Dysregulation of the complement system has been shown in genetic, histological, and preclinical studies to be associated with inflammation and the formation of a protein complex, which directly damages photoreceptor and retinal pigment epithelium (RPE) cells, leading to atrophy.1
Pegcetacoplan binds to, and inhibits, complement factors C3 and C3b, whereas avacincaptad pegol acts further downstream at C5. Both treatments are administered via intravitreal injection.
The efficacy of pegcetacoplan was shown in the DERBY and OAKS Phase 3 clinical trials.2 In these studies, pegcetacoplan monthly and every-other-month slowed GA lesion growth by 22% and 18% in OAKS, and by 19% and 16% in DERBY, respectively over two years, compared to eyes that were treated with sham injections where GA progressed at a normal rate. Meanwhile, the efficacy of avacincaptad pegol was shown in the GATHER2 Phase 3 trial.3 Unlike in OAKS and DERBY, which included GA lesions that could have foveal involvement (sub-foveal), GATHER2 only included patients where GA did not involve the fovea (non-subfoveal). In this study, eyes that received avacincaptad pegol 2 mg monthly showed reductions in GA growth rate by 14% over 24 months compared to eyes treated with sham injections.4 Meanwhile, eyes initially treated monthly who switched to every-other-month dosing in the second year showed a 19% reduction in GA lesion growth by two years.
Long-term efficacy of pegcetacoplan has been studied in the GALE three-year open-label extension study where all patients received pegcetacoplan.5 Because all patients in the sham arm crossed over to active treatment, a projected sham was used to model GA progression as the comparator without treatment. The five-year results showed further reductions in rate of GA growth, with patients treated with pegcetacoplan every-other-month for 60 months preserving a total of 3.4 mm2 of retinal tissue, equivalent to ~1.5 disc areas. Meanwhile, patients who originally received sham treatment and crossed over to receive pegcetacoplan after 24 months still preserved retinal tissue, however the treatment effect was only approximately half of that experienced by those who started earlier.6 Furthermore, fellow eyes of patients who had bilateral GA showed continuous linear lesion growth without treatment. This supports the long-term efficacy of GA therapies and suggests that earlier treatment leads to improved anatomical outcomes.

Dr Cheryl Au

Dr Long Phan

Prof Andrew Chang AM
These anatomic outcomes have also been associated with meaningful functional benefits for patients receiving pegcetacoplan. While treatment has shown limited impact on visual acuity, visual acuity is known to correlate poorly with geographic atrophy. However, several benefits to alternative aspects of visual function, such as retinal sensitivity, low-luminance acuity, and driving eligibility have all been observed, which ultimately can lead to improved vision-related quality of life for GA patients receiving therapy.7-9
PATIENT ELIGIBILITY FOR TREATMENT IN AUSTRALIA
GA therapy slows the rate of progression of GA and preserves retinal tissue.
However, because current GA therapy does not reverse GA growth or restore vision, patients who still have central vision, but whose fovea is threatened by GA growth, would benefit most from therapy (Figures 2 and 3). This forms the basis of the TGA approval criteria for pegcetacoplan in Australia. Because only a minimal added benefit for a higher dosing frequency was shown in OAKS and DERBY,2 an every-other-month regime was approved for the treatment of GA secondary to AMD. Similarly, avacincaptad pegol is approved for every-other-month treatment, following every month treatment for the first year.
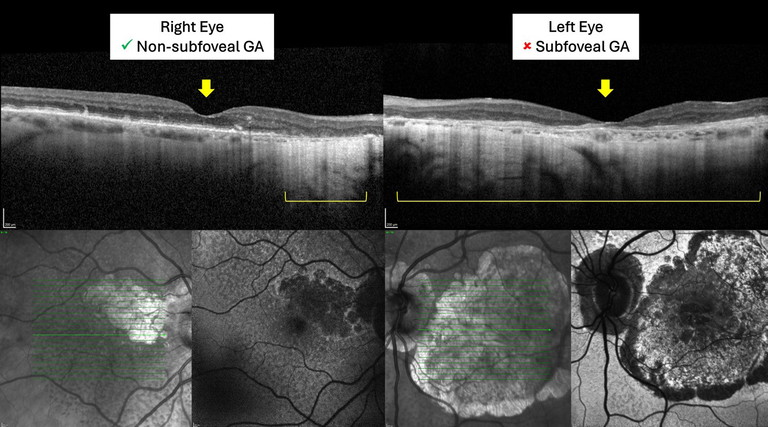
Figure 2. OCT, NIR, and FAF images of a 90-year-old patient with bilateral GA. Yellow arrows show the centre point of the fovea, yellow brackets show hyper-transmission defects corresponding to GA on OCT. Visual acuity is 6/7.5 in the right eye and 6/120 in the left eye. Images from previous visits showed progression of GA towards the fovea. Because the GA in the right eye was non-subfoveal, treatment to slow progression was discussed in order to preserve vision.
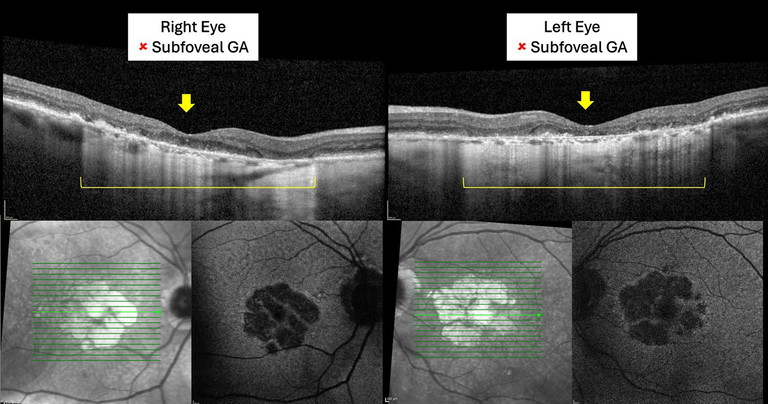
Figure 3. A patient with bilateral GA. Visual acuity is 6/90 in both eyes. Because the GA is subfoveal in both eyes, this patient would not be suitable for currently approved GA therapies under the proposed criteria.
While neither pegcetacoplan nor avacincaptad pegol are currently listed on the PBS, the positive PBAC recommendation of Syfovre provides clinicians insight into the proposed PBS criteria, which will likely require patients to have:
• GA secondary to AMD,
• AND the condition must be diagnosed by optical coherence tomography or fundus autofluorescence,
• AND the patient must have non-subfoveal GA lesion(s), distanced from the centre point of the fovea by at least 1 micrometre (μm) in the treated eye,
• AND the patient must have subfoveal GA lesion(s) involving the foveal centre point in the non-treated eye causing vision impairment (i.e. the fovea is no longer intact),
• AND the patient must have central vision in the treated eye threatened by growth of GA lesion(s), defined as either:
i. Located within the 750 µm radius of the foveal centre, or
ii. Located beyond 750 µm from the foveal centre, where historical imaging shows lesion progression toward the foveal centre, and is located within the macula (defined as the 5.5 mm diameter area).
• AND the treatment must be the sole intravitreal injection therapy for GA.
Syfovre is contraindicated in patients with hypersensitivity to pegcetacoplan or to any of the excipients, ocular or periocular infections, and/or active intraocular inflammation.
Clinicians need to examine patients before starting treatment and to monitor them carefully when on therapy.
DIAGNOSIS OF GEOGRAPHIC ATROPHY
Clinicians first need to verify the presence of GA secondary to AMD, by excluding other diseases that also present with retinal atrophy. Inherited retinal diseases are caused by gene defects, and some of them have macular involvement, which can look very similar to GA e.g., Stargardt disease and adult onset vitelliform macular dystrophy. A detailed medical and family history, combined with multimodal imaging will aid in the differential diagnosis of patients with GA.
“Because current GA therapy does not reverse GA growth or restore vision, patients who still have central vision, but whose fovea is threatened by GA growth, would benefit most from therapy”
Multimodal imaging used to investigate GA includes colour fundus photography, fundus autofluorescence (FAF) imaging, optical coherence tomography (OCT), and near infrared reflectance (NIR) imaging. Each technique highlights different structural features of the disease, offering valuable complementary information to document progression rates and to understand the natural history of GA. However, given that documented GA progression may be a requirement for accessing treatment, it is important that clinicians repeat imaging protocols from previous visits for each patient, in order to reliably compare images obtained using the same modality and settings over time.
Colour fundus imaging or fundus examination remains the first approach to detect GA. GA is characterised by a sharply demarcated round or oval lesion with a regular or multi-lobed appearance, consisting of one or multiple foci. In the absence of RPE, large choroidal vessels are visible.
The area of atrophy on colour fundus imaging corresponds to decreased signal or hypo-autofluorescence on FAF. Perilesional or junctional FAF patterns have been extensively used as prognostic indicators of GA growth. The classification system is based on patterns of hyper-autofluorescence at the rim or junction of the GA lesion, and includes focal, banded, diffuse, and trickling. Studies have shown that banded, diffuse, and diffusetrickling phenotypes are associated with more rapid lesion expansion.10
OCT is now the gold standard for diagnosing and monitoring retinal conditions, and GA is no exception. The Classification of Atrophy (CAM) consensus group defined GA using the term complete RPE and outer retina atrophy (cRORA), which is characterised by signal hypertransmission ≥250 μm, as well as complete loss of RPE and photoreceptors ≥250 μm, in the absence of RPE tear or scrolled RPE. Incomplete RPE and outer retina atrophy (iRORA) is defined by the same features as cRORA however the region of choroidal hypertransmission and corresponding region of RPE attenuation or disruption is <250 μm. Longitudinal studies confirmed the concept of progression from iRORA to cRORA.11
NIR is also readily available as part of OCT acquisition, where GA appears as a hyperreflective lesion with sharply demarcated borders. Both NIR and FAF can be used to measure the area of a GA lesion.
Modern imaging techniques have allowed the investigation of subtle changes associated with GA at the photoreceptor level. Studies have shown that regions with ellipsoid zone (EZ) loss or photoreceptor thinning are associated with future GA progression, making these features valuable for predicting disease progression and counselling patients.12 Artificial intelligence-based software, such as the RetInSight GA Monitor (RetInSight GmbH, Vienna) have been developed to allow routine quantification and visualisation of these changes. RetInSight GA Monitor has been approved by the TGA as of August 2025 as a Class IIa medical device, allowing it to be used as a clinical support tool.
RISK FACTORS FOR PROGRESSION
Not all GA progresses at the same rate.13 When GA becomes subfoveal, central vision is severely impaired with rapid loss of visual function (Figure 4). Identification of clinical and lesion characteristics that are more susceptible to faster growth is vital in GA treatment and patient counselling. The following characteristics are correlated to faster GA expansion:14
Clinical Features
• Bilateral GA involvement,
• Patients with relatively preserved visual function at baseline may paradoxically be at risk for faster GA progression,
• In the OAKS and DERBY trials, eyes with BCVA >60 letters (Snellen 6/19 demonstrated faster lesion growth than those with poorer BCVA (p=0.0020),2
• Reticular pseudodrusen or subretinal drusenoid deposits are highly linked to GA progression,15
• Drusen volume of 0.03 mm3 or more,15
• Intraretinal hyperreflective foci on OCT, and
• Features of iRORA.11
Lesion Features
• Larger lesion size. In an observational study,17 lesions measuring <1.3 mm2 , 1.3 to 8.3 mm2 , and >8.3 mm2 had progression rates of 0.8 mm2 /year, 2.1 mm2 /year, and 3.0 mm2 /year, respectively.
• Lesion location. In the OAKS and DERBY trials, non-subfoveal or extrafoveal lesions exhibited significantly faster growth over 24 months compared with subfoveal lesions (p<0.0001).14
• Lesion focality. In the OAKS and DERBY trials, eyes with multifocal GA lesions showed significantly faster growth at 24 months compared with unifocal lesions (0.33 vs 0.28 mm/year; p=0.0003).14
• Lesion circularity. A lower circularity index (<0.2) was associated with faster growth rates, while more circular lesions (≥0.4) enlarged at a slower rate (p<0.0001).14
• FAF pattern. Banded, diffuse and diffusetrickling junction or perilesional FAF patterns are associated with more rapid lesion growth.18
CONCLUSION
After not having any treatments for GA secondary to AMD beyond recommending vitamins and supplements or referrals for low vision services, the TGA approval of both pegcetacoplan and avacincaptad pegol in Australia is a milestone for clinicians and offers hope for patients and family members affected by GA.
The challenge now lies in identifying eligible patients that will derive maximal benefit from these therapies to maintain their visual function and quality of life. The positive PBAC recommendation for Syfovre is an important initial milestone towards improved access to GA treatment for Australian patients.
Patient education, counselling, and appropriate referral are required to confirm diagnosis by multimodal imaging, determine eligibility for GA treatment, manage expectations, and promote adherence.
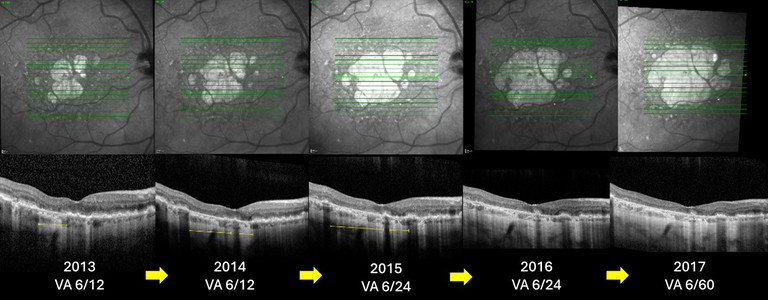
Figure 4. Progression of GA on NIR imaging and OCT. The patient developed significant vision impairment following expansion over the centre point of the fovea.
To earn your CPD hours from this article, scan the QR code or visit mieducation.com/new-approved-treatments-forgeographic-atrophy-what-youneed-to-know.
Dr Cheryl Au BMedSci MPH, MBBS (Hon I) FRANZCO is a comprehensive ophthalmologist and cataract surgeon, who subspecialises in medical retina. She was the Sarks’ research fellow at St Vincent’s Hospital, where she undertook research on age-related macular degeneration based on the Sarks’ clinicopathological archive, the world’s largest collection of clinically annotated eyes. Dr Au is a staff specialist at Sydney Eye Hospital, and is also an adjunct lecturer at the University of Notre Dame.
Dr Long Phan BPharm MOrth PhD is an orthoptist and researcher. He conducts investigator-initiated research at CUREOS, a clinical research network conducting ophthalmology trials in retinal disease. He has been involved in several pharmaceutical sponsored trials as a coordinator and examiner. He has also lectured research subjects within the Master of Orthoptics program at the University of Technology Sydney. Professor Andrew Chang AM MBBS (Hons) PhD FRANZCO FRACS is a vitreoretinal surgeon and ophthalmologist. He holds academic appointments of Conjoint Professor Department of Surgery UNSW and Clinical Associate Professor at the University of Sydney.
He is a consultant ophthalmologist and the Head of Ophthalmology at the Sydney Eye Hospital. He is the Medical Director of Sydney Retina Clinic. Prof Chang is clinician advisor to the Department of Health Australia and to the Therapeutic Goods Administration on new technologies in ophthalmology. In 2023 he was awarded as a Member of the Order of Australia (AM), in recognition of his contributions to ophthalmology and retinal surgery.

“Given that documented GA progression may be a requirement for accessing treatment, it is important that clinicians repeat imaging protocols from previous visits for each patient, in order to reliably compare images”
References available at mivision.com.au.