miophthalmology
Laser Keratorefractive Surgery: To Do or Not? That is the Question
WRITER Associate Professor Rob Paul
While laser refractive surgery is an extraordinarily common procedure, it is not suitable for everyone. Associate Professor Rob Paul reminds us that while medical professionals pledge to “do no harm”, this does not mean “do surgery on all patients”. Indeed, a very good surgeon knows when to operate – and when not to.
As the Medical Director of Perth’s most prominent and highest volume laser refractive centre, I pride myself on using the most advanced technology and employing the most highly qualified surgeons. We need the best lasers and technology to maximise patient outcomes and to attract quality staff. My mantra also includes maintaining high ethical standards. It’s for this latter reason that I abide by the concept that a very good surgeon knows when to, and when not to operate.
The conundrum that I – and all ophthalmic surgeons – face is that after investing a large amount of capital and effort in establishing a successful practice, the drive to remain profitable is paramount.
To echo and transform the question posed by Shakespeare’s Hamlet, today’s refractive surgeons may be asking themselves “to do, or not to do” when it comes to laser refractive surgery. The question can be avoided by screening patients appropriately.
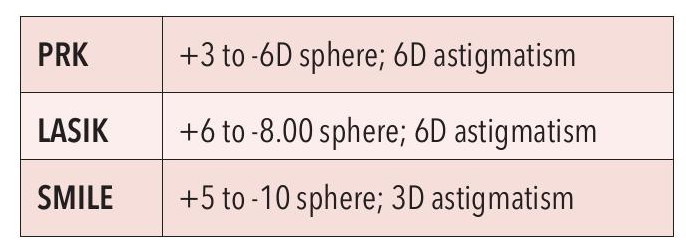
Only then can we categorise those situations where laser refractive surgery – laser-assisted in situ keratomileusis (LASIK), small incision lenticule extraction (SMILE) and photorefractive keratectomy (PRK), may not be the best option.
CORNEAL ISSUES: TOPOGRAPHY, TOMOGRAPHY, AND BIOMECHANICS
Corneal ectasia is one of the most devastating complications after LASIK and other refractive surgical procedures. Post-LASIK ectasia is considered in patients who develop increasing myopia (with or without increasing astigmatism), loss of uncorrected visual acuity – and often loss of best corrected visual acuity (BCVA) – with keratometric steepening, posterior corneal elevation (with or without central and paracentral corneal thinning), and topographic evidence of asymmetric inferior corneal steepening after a LASIK procedure.1 Ectatic changes can occur as early as one week2 or can be delayed for years after LASIK.3,4 The actual incidence of ectasia is undetermined, although incidence rates of 0.04%5 to 0.2%6 to 0.6%7 have been reported. Screening against ectasia is absolutely vital.
Risk Factors The Ectasia Risk Score System, designed by Randleman and colleagues,8 is a controversial screening tool carefully developed following an evidence-based review of a large series of LASIK ectasia cases. The Ectasia Risk Score System scale may help to identify high-risk patients preoperatively. It is described as controversial in its utility because diagnostic methods of screening for preoperative ectasia have changed dramatically over the past 10 years, and now include possibly more sensitive techniques of corneal ‘tomography’ (such as Pentacam, MS39, Gallei), which can measure posterior corneal curvature, rather than placido-disc based ‘topography’, which only measures anterior corneal curvatures. In the Randleman et al. score system, the most common risk factors, in order of significance, include:
Abnormal preoperative topography.
Abnormal topography compromises keratoconus, pellucid marginal corneal degeneration, or sub-clinical (forme fruste) keratoconus with an inferior-superior (I-S) value of 1.4 or more9 and is a significant factor with high relative risk. The I-S index compares the power difference between five points in the inferior hemisphere to the superior located 3 mm from the corneal apex.
The axial map placido disc-based topography pattern classification using in this score system consists of:
• Normal/symmetrical, including round, oval, or symmetric bowtie patterns.
• Suspicious, including the following asymmetric patterns:
Asymmetric bowtie. 1. Asymmetric steepening in any direction less than 1.0 diopter (D); 2. No skewed radial axis.
Inferior steep/skewed radial axis.
1. Significant skewed radial axis with or without inferior steepening; 2. One diopter or more of inferior steepening in some areas but an I-S value of less than 1.4.9,10
• Abnormal includes keratoconus, pellucid marginal corneal degeneration, or subclinical (forme fruste) keratoconus with an I-S value of 1.4 or more.9

“Excluding a patient from laser surgery is not necessarily detrimental to a practice’s success; it demonstrates honesty to the patient and the referrer”

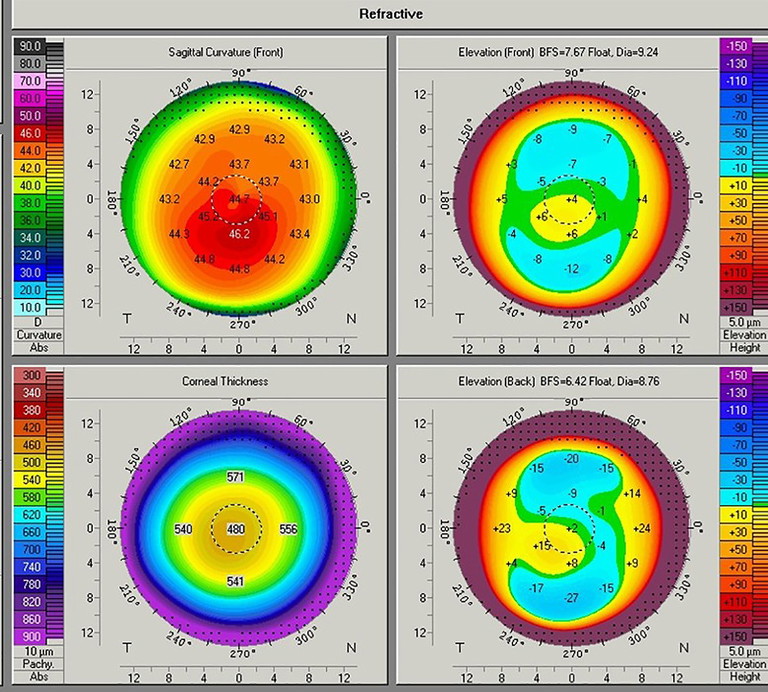
Figure 1A. Pentacam analysis showing early keratoconus.
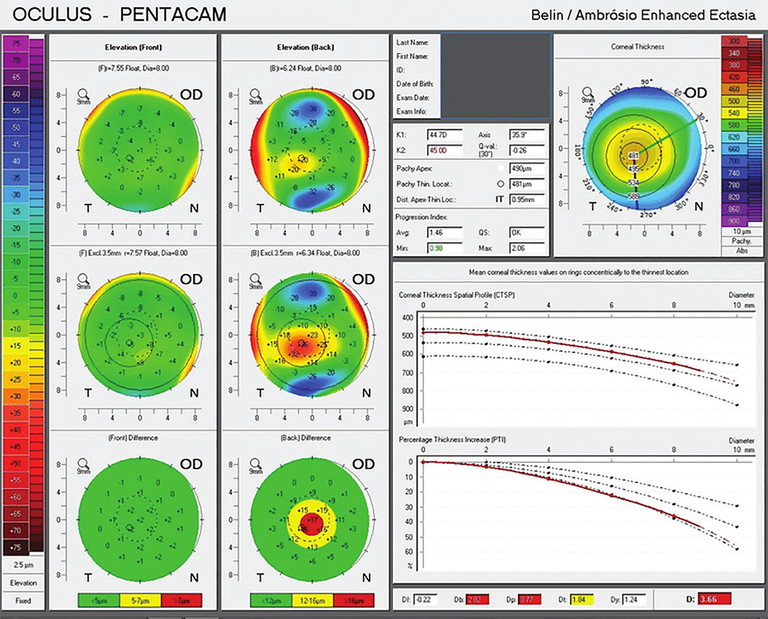
Figure 1B. The Beilin /Ambrosio Display (BAD) of a patient with mild keratoconus. Three of the five parameters – Db (back elevation), Dp (pachymetric progression) and Dt (corneal thinnest point) – are abnormal, lying outside normative standard deviations.
Low Residual Stromal Bed Thickness
The original standard for minimum stromal bed thickness after LASIK was 250 μm (preop central corneal thickness minus flap thickness minus ablation depth equals residual stromal bed (RSB)). This figure came from research comparing the tangential elastic modulus of the normal cornea with that of the keratoconic cornea. It suggested the normal cornea is 2.1 times stronger than the keratoconic cornea. The 250 μm is derived from dividing an average corneal thickness of 520 μm by 2.1 (=250). Modern ideology dictates a safety margin higher than this for LASIK of 270–300 μm. Interestingly, SMILE or KLEX treatments adhere to the 250 μm rule as they are considered to weaken the cornea less.
RSB thickness is especially important after LASIK because stress-strain analysis11 indicates greater strength in the anterior 40% relative to posterior 60% of stroma, and LASIK reduces anterior corneal structural integrity.12
Ectasia increases reciprocally, relative to RSB thickness, and a RSB of <300 μm has been correlated with increased risk of ectasia.13 The use of thin flap LASIK or thinner caps in SMILE/KLEX allows for a greater RSB buffer.
Younger Age
Younger age may be a significant risk factor for ectasia in patients without other risk factors. One hypothesis is that some of these individuals would have developed delayed onset sub-clinical or frank keratoconus, even without LASIK.
Low Preoperative Corneal Thickness
Corneal thickness, degree of myopia, and RSB are related, and RSB thickness is the most significant predictor of ectasia among them. Traditionally, a corneal thickness below 500 μm was considered ineligible for LASIK.
Modern thinking has shifted as a 490 μm cornea in a 50-year-old is not as high risk as in a 20-year-old. This is especially the case if other parameters, such as the corneal vertex displacement, anterior, and posterior corneal elevation, are considered normal. I would not do LASIK / SMILE on a cornea under 480 μm and I would not do PRK under 440 μm under any circumstance. For PRK, the central corneal thickness postoperatively should be greater than 410 μm (350 RSB).
High Myopia
Despite the early reported cases of ectasia for extreme myopia (more than 12D), post-LASIK ectasia has been reported in numerous patients with low myopia14 and even hyperopia.15
The Ectasia Risk Score System is a cumulative score system. Risk categories based on points are:
• 0–2 points equals low risk,
• Three points equals moderate risk,
• Four points equals high risk.
The score system may be summarised as set out in Table 1.8
Other risk factors include eye rubbing, family history of keratoconus, refractive instability, and BCVA less than 20/20 preoperatively, male gender. These factors should all be considered, especially in borderline cases.
One significant criticism of the Ectasia Risk Score System is that any individual less than 22 years old is automatically classified as at least ‘moderate risk’, despite the low incidence of ectasia in this age group.
Percent Tissue Altered
Percent Tissue Altered (PTA) is an additional significant risk factor for post-LASIK ectasia and may be considered in addition to residual stromal bed when determining the safety of an excimer laser treatment. PTA is equal to the flap thickness (FT) plus the ablation depth (AD) divided by the pre-operative thinnest central corneal thickness (CCT).16
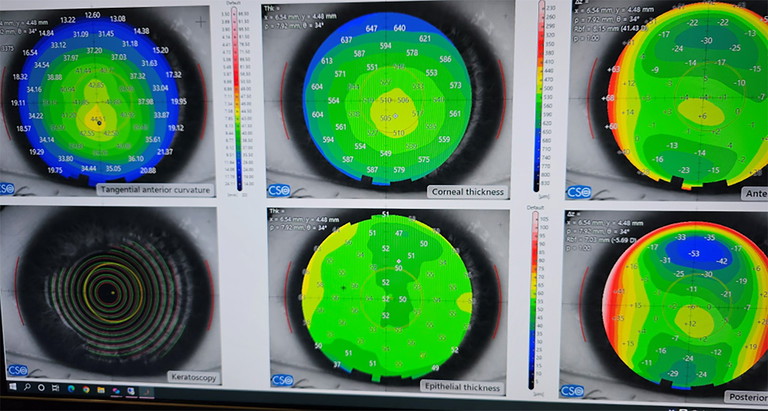
Figure 2A. Epithelial thickness is sometimes thinner over the apex of the cone and is diagnostic of keratoconus.
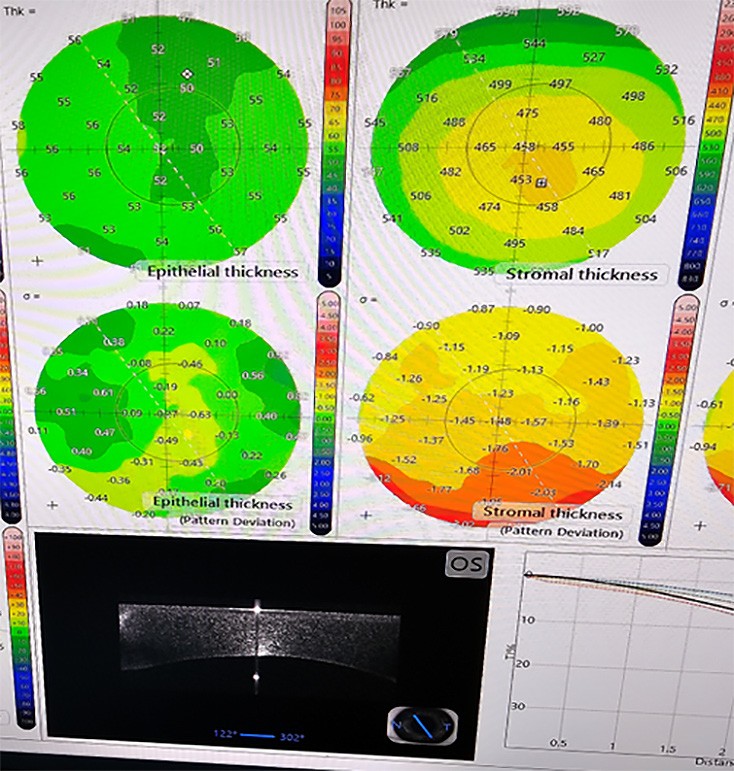
Figure 2B. MS39 keratoconus screening takes into account epithelium and OCT imaging.
A PTA of 40% is an indicator of a higher risk for ectasia.17
PTA = (FT + AD)/CCT]. Some studies have indicated that PTA is one of the most predictive risk factors for corneal ectasia, even in eyes with a normal pre-operative corneal topography.18
Central Keratometry
Values below 47.2D are considered normal; between 47.2 and 48.7D are probable keratoconus cases; and above 48.7D are clinical keratoconus.19
Modern Tomography/Biomechanics
Pentacam and/or Corvis corneal indices, such as Belin/Ambrosio enhanced ectasia display (BAD) and the Corvis Biomechanical Index (CBI), should be considered prime screening tools to exclude a patient from laser refractive surgery. The Corvis uses a high-speed Scheimpflug camera to assess the cornea’s response to a puff of air. The Corvis tomographic biomechanical index (TBI) combines Pentacam data with corneal deformation and is another useful detection tool. These investigations should be considered alongside the Randelman’s score, with the latter an adjunct tool. The BAD software combines anterior and posterior elevation data with pachymetric data to produce a three-dimensional tomographic representation of the cornea. This can lead to early detection of keratoconus and post-laser ectasia that can be missed with classic placido and corneal thickness measurements.
Systems, such as the MS39, incorporate OCT analysis of the posterior cornea into keratoconus screening algorithms and include epithelial thickness. This is extremely useful when diagnosing at-risk corneas.
In cases of suspicious or form fruste keratoconus, PRK, LASIK and SMILE can be combined with corneal crosslinking (CXL). More significant cases may be amenable to implantable collamer lenses (ICL), or implantable phakic contact lenses (IPCL), or even a corneal allogenic intrastromal ring segment (CAIRS) procedure.
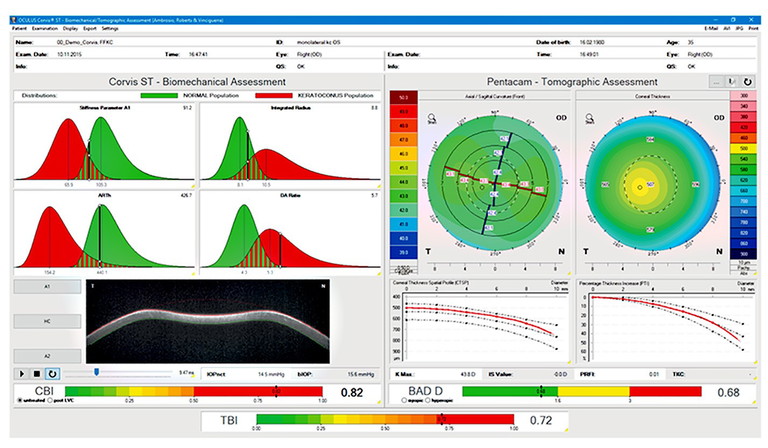
Figure 3. Corvis uses corneal deformation combined with Pentacam analysis to detect early keratoconus.
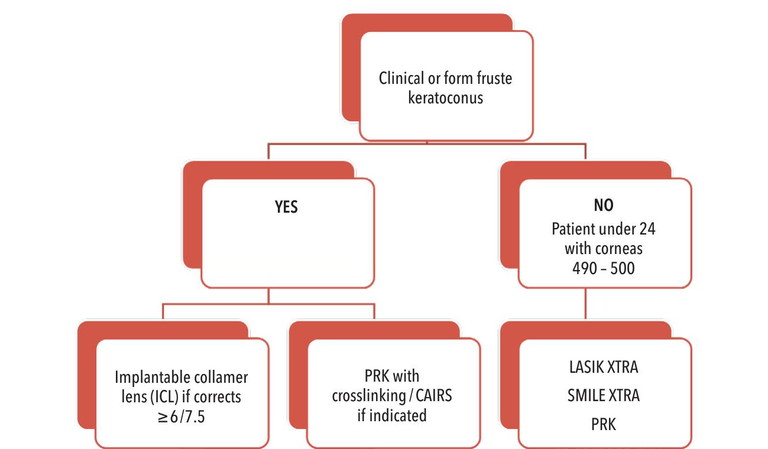
Figure 4. Flowchart for laser refractive surgery as it relates to ectasia risk.
Assuming my patient is not a candidate for a corneal transplant or hard contact lens wear, my flowchart for laser refractive surgery, as it relates to ectasia risk, is summarised in Figure 4.
If I am unsure about performing laser because of the ectasia risk, I often ask myself, ‘what if the patient did have this outcome?’. If I retrospectively reviewed the scans and wished I’d never done it, then my answer becomes easy. Unfortunately, it’s not always this easy. Sometimes ectasia can strike without pre-existing evidence.
REFRACTION
An unstable refraction – defined as greater than a 0.5D change over 12 months – is more common before the eye is fully grown. This occurs around 20 to 21 years old. If a patient is over 18 but the error is mild and stable, laser can be considered, and overcorrection of the myopic error is employed to cater for eye growth. A cycloplegic refraction is important, especially for hyperopic young patients, to unmask latent hyperopia. Pregnant patients should not have surgery after the first trimester; breastfeeding patients should not have surgery within the first three months of giving birth. Orthokeratology (OK) patients should be out of lenses for at least six months prior to refraction.
My refractive limits are as follows; for anything outside this range I perform ICL surgery.
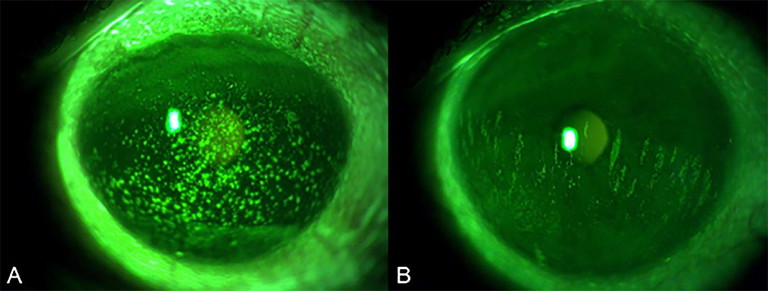
Figure 5A and 5B. Aqueous deficiency related dry eye.20
Figure 6. Fuchs’ corneal dystrophy.
CORNEAL PATHOLOGY
Significant dry eye (especially neurotrophic), epithelial basement membrane dystrophy, Fuchs’ endothelial dystrophy, and corneal scars from any cause (trauma/ herpes simplex virus/ keratitis) are contraindications to LASIK and SMILE. These patients are better served by performing PRK (providing the cornea is not so irregular or thin that the treatment becomes unpredictable). Transepithelial (with or without topography guided) PRK and Mitomycin C are the best options.
LENS ISSUES AND GLAUCOMA
Cataract and subluxed lenses (Marfan’s syndrome/trauma) can lead to unstable refractions as well as higher-order aberrations. Lens-based surgery is advisable for these patients as they are not suitable for laser.
In moderate to severe glaucoma, there are several risks. The applanation pressure of planar interface femtosecond lasers can reach 100–200 mmHg, placing the optic nerve at risk. Curved interfaces, such as the Visumax 800, are lower pressure and safer systems. Treating large refractive errors, especially myopia, can reduce contrast sensitivity, thereby affecting the accuracy of visual field testing. Additionally, treating large refractive errors can create falsely low intraocular pressure (IOP) readings, which may lead to complacency when managing glaucoma patients. To compensate, the IOP must always be checked in the periphery of post-laser glaucoma individuals. Pressure-induced stromal keratitis can occur post laser refractive surgery (in LASIK especially), which leads to a myopic shift and falsely lower IOP.
RETINAL ISSUES
If age-related macular degeneration, myopic macular degeneration or retinitis pigmentosa are present, then laser is not recommended. The caveat to this is that high myopes under 60 years of age may benefit more from laser than a lens refractive procedure, as there is much less risk of retinal detachment.20
BINOCULAR VISION ISSUES
Strabismus, when accompanied by significant amblyopia, needs careful consideration. Patient expectations must be realistic, and one must be aware that abnormal retinal correspondence can lead to diplopia postlaser if the eyes do not alternate with fixation. Unmasking a diplopia previously hidden by glasses can be cosmetically unacceptable and, if not a refractive /accommodative esotropia, one should suggest strabismus repair prior to laser. If a patient’s preop glasses incorporate prisms, the refractive surgeon should always be aware of the risk this poses in terms of diplopia postlaser refractive surgery, and the ongoing need to wear glasses. Nystagmus is not a contraindication for laser refractive surgery if the excimer laser has a modern tracking system including cyclotorsion. However, SMILE surgery may pose a challenge in the manual dissection phase.
PATIENT ISSUES
If a patient is involved in contact sports, LASIK may not be the best option. SMILE or PRK may be better to avoid flap dislocation. I personally have performed LASIK on high-level AFL players and cricketers without ill effects, however rugby or mixed martial arts sportspeople would not be good LASIK candidates.
Other patients that I do not consider as good candidates for LASIK include those with autoimmune diseases, such as Sjögren’s syndrome, rheumatoid arthritis, and ocular cicatricial pemphigoid. Patients to treat with caution include those on certain medications such as steroids (due to unstable refraction and cataract development risk), Roaccutane (due to potential scarring in PRK, dry eye), and antidepressants or antihistamines (both due to dry eye). I suggest waiting until they come off these medications, noting that for Roaccutane, the effects can persist for a year after cessation.
As with any elective surgery, treatment often includes expectation management. Overdemanding, litigious, and aggressive patients are contraindications in certain situations – there’s no crystal ball to distinguish between an anxious patient and one who will never be satisfied. Personally, I’m not perturbed by performing surgery on demanding patients, but I ensure they are fully aware of every likely and unforeseen complication that can arise.
Informed consent is essential in every case. That’s not to say you should never say no to someone based on their personality. If they are abusive to you or your staff, constantly interrupt, or are untruthful, that behaviour should be seen as a red flag.
SIDE EFFECTS
LASIK patient satisfaction rates vary from 82 to 98%.21-24
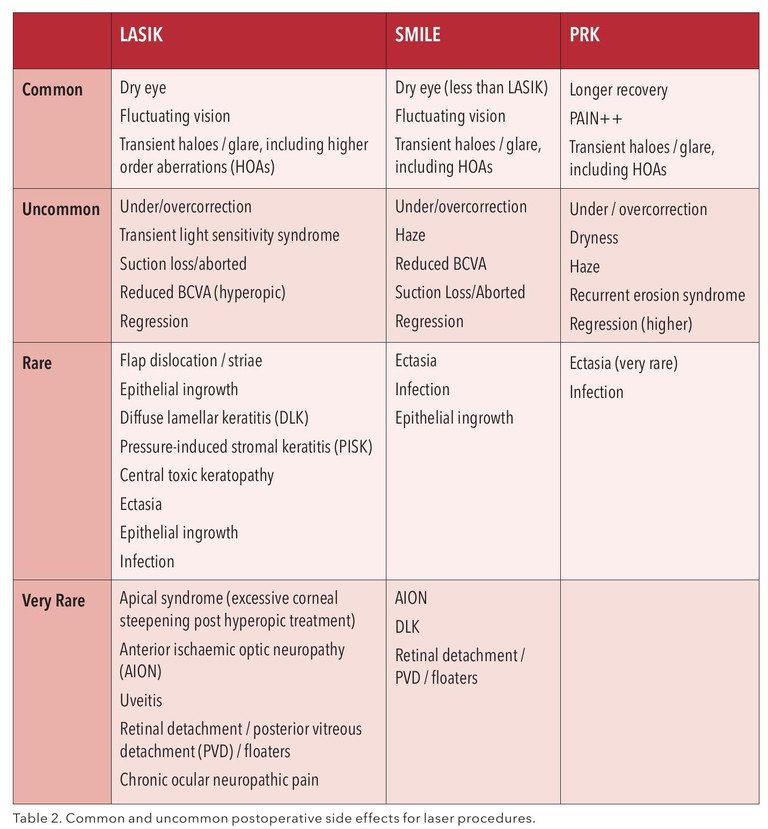
Common and uncommon postoperative side effects for laser procedures are listed in Table 2.
It must be remembered that serious risks, like infection following a laser procedure, are extremely low, being 0.035% following LASIK in an analysis of more than 100,000 patients.25
Beliefs that a patient may eventually go completely blind after laser, that it’s extremely painful, or wears off quickly, are unfounded in the modern era.
The evolution and success of presbyopic LASIK treatments, such as Presbyond and Presbymax, which manipulate spherical aberration and allow for increased binocular depth of focus, have been revolutionary. This means that reading problems in pre and presbyopic patients can now be addressed with laser. While laser monovision is effective for some patients, the future of presbyopic laser treatments will head more in the direction of controlling spherical aberration.
SUMMARY
Laser refractive surgery has helped over 40 million people since its inception.26,27 Its success, however, needs to be calibrated with correct decision making by surgeons. Excluding a patient from laser surgery is not necessarily detrimental to a practice’s success; it demonstrates honesty to the patient and the referrer, sometimes leading to more referrals. Alternative options such as ICL, IPCL, CAIRS or lens-based procedures may be possible. In contrast, a bad outcome in someone who should not have had surgery can, as in Hamlet, end in tragedy.
Complications will occur despite the best screening processes. As a young service registrar in cardiothoracic surgery, I was once told: “If you don’t want complications, don’t operate”. It thus becomes a question of minimising risk and always considering patient welfare above all else.
“Laser refractive surgery has helped over 40 million people...”
To earn your CPD hours from this article, scan the code or visit mieducation.com/laser-keratorefra-ctive-surgery-to-do-ornot-to-do-that-is-the-question.
Associate Professor Rob Paul MBBS (UWA) FRANZCO is Medical Director and lead surgeon at WA Laser Eye Centre. He is recognised as Western Australia’s highest volume laser surgeon and the State’s first clinically fellowship-trained surgeon in corneal and refractive surgery (University of Toronto). His special interests are in cataract, corneal, and laser refractive surgery.

Assoc Prof Paul has held several positions with the Royal Australian and New Zealand College of Ophthalmologists in WA, he is Adjunct Senior Lecturer at Curtin Medical School and Adjunct Associate Professor at the University of Western Australia School of Optometry. He is well-published in peer-reviewed journals and mentors/trains students, ophthalmologists, and refractive surgeons.
Financial disclosures: Assoc Prof Paul is a paid Consultant for ZEISS, Johnson and Johnson, Alcon, and Bausch and Lomb.
References
1. Binder PS, Lindstrom RL, Rabinowitz Y, et al. Keratoconus and corneal ectasia after LASIK [letter]. J Cataract Refract Surg. 2005; 31:2035-2038. doi: 10.1016/j.jcrs.2005.12.002.
2. Rao SN, Epstein RJ. Early onset ectasia following laser in situ keratomileusus: case report and literature review. J Refract Surg. 2002 Mar-Apr;18(2):177-84. doi: 10.3928/1081-597X-20020301-13.
3. Geggel HS, Talley AR. Delayed onset keratectasia following laser in situ keratomileusis. J Cataract Refract Surg. 1999 Apr;25(4):582-6. doi: 10.1016/s0886-3350(99)80060-1.
4. Lifshitz T, Levy J, Klemperer I, Levinger S. Late bilateral keratectasia after LASIK in a low myopic patient. J Refract Surg. 2005 Sep-Oct;21(5):494-6. doi: 10.3928/1081-597X-20050901-12.
5. Randleman JB, Russell B, Ward MA, et al. Risk factors and prognosis for corneal ectasia after LASIK. Ophthalmology. 2003; 110:267-275. doi: 10.1016/S0161-6420(02)01727-X.
6. Rad AS, Jabbarvand M, Saifi N. Progressive keratectasia after laser in situ keratomileusis. J Refract Surg. 2004; 20:S718–S722. doi: 10.3928/1081-597X-20040903-18.
7. Pallikaris IG, Kymionis GD, Astyrakakis NI. Corneal ectasia induced by laser in situ keratomileusis. J Cataract Refract Surg. 2001; 27:1796-1802. doi: 10.1016/s0886-3350(01)01090-2.
8. Randleman JB, Russell B, Ward MA, Thompson KP, Stulting RD. Risk factors and prognosis for corneal ectasia after LASIK. Ophthalmology. 2003 Feb;110(2):267-75. doi: 10.1016/S0161-6420(02)01727-X.
9. Rabinowitz YS, McDonnell PJ. Computer-assisted corneal topography in keratoconus. Refract Corneal Surg. 1989 Nov-Dec;5(6):400-8. PMID: 2488838.
10. Rabinowitz YS. Videokeratographic indices to aid in screening for keratoconus. J Refract Surg. 1995 Sep-Oct;11(5):371-9. doi: 10.3928/1081-597X-19950901-14.
11. Kohlhaas M, Spoerl E, Pillunat LE, et al. Biomechanical evidence of the distribution of cross-links in corneas treated with riboflavin and ultraviolet A light. J Cataract Refract Surg. 2006 Feb;32(2):279-83. doi: 10.1016/j.jcrs.2005.12.092.
12. Randleman JB, Dawson DG, Edelhauser HF, et al. Depth-dependent cohesive tensile strength in human donor corneas: implications for refractive surgery. J Refract Surg. 2008 Jan;24(1):S85-9. doi: 10.3928/1081597X-20080101-15. .
13. Santhiago MR, Smadja D, Randleman JB, et al. Association between the percent tissue altered and post-laser in situ keratomileusis ectasia in eyes with normal preoperative topography. Am J Ophthalmol. 2014 Jul;158(1):87-95.e1. doi: 10.1016/j.ajo.2014.04.002.
14. Amoils SP, Deist MB, Gous P, Amoils PM. Iatrogenic keratectasia after laser in situ keratomileusis for less than -4.0 to -7.0 diopters of myopia. J Cataract Refract Surg 2000;26: 967–77. doi; 10.1016/S0886-3350(00)00434-X.
15. Randleman JB, Banning CS, Stulting RD. Corneal ectasia after hyperopic LASIK. J Refract Surg. 2007 Jan;23(1):98-102. doi: 10.3928/1081-597X-20070101-17.
16. Santhiago MR, Smadja D, Randleman JB, et al. Association between the percent tissue altered and post-laser in situ keratomileusis ectasia in eyes with normal preoperative topography. Am J Ophthalmol. 2014 Jul;158(1):87-95.e1. doi: 10.1016/j.ajo.2014.04.002.
17. Santhiago MR. Percent tissue altered and corneal ectasia. Curr Opin Ophthalmol. 2016 Jul;27(4):311-5. doi: 10.1097/ICU.0000000000000276.
18. Feldman BH, Kozak A, Severin Pouly MD, et al. Ectasia after lasik. Eye Wiki . Available at: eyewiki.org/Ectasia_After_LASIK [accessed Nov 2025].
19. Maeda N, Klyce SD, Smolek MK, Thompson HW. Automated keratoconus screening with corneal topography analysis. Invest Ophthalmol Vis Sci. 1994 May;35(6):2749-57. PMID: 8188468.
20. Pellegrini M, Bernabei F, Giannaccare G, et al. Assessment of corneal fluorescein staining in different dry eye subtypes using digital image analysis. Transl Vis Sci Technol. 2019 Dec 12;8(6):34. doi: 10.1167/tvst.8.6.34.
21. Stephenson,M. A review of refractive lens exchange. Review of Ophthalmology. Nov 2022. Available at reviewofophthalmology.com/article/a-review-of-refractive-lens-exchange [accessed Nov 2025].
22. Tahzib NG, Bootsma SJ, Nuijts RM, et al. Functional outcomes and patient satisfaction after laser in situ keratomileusis for correction of myopia. J Cataract Refract Surg. 2005 Oct;31(10):1943-51. doi: 10.1016/j.jcrs.2005.08.022.
23. Bailey MD, Mitchell GL, Zadnik K, et al. Patient satisfaction and visual symptoms after laser in situ keratomileusis. Ophthalmology. 2003 Jul;110(7):1371-8. doi: 10.1016/S0161-6420(03)00455-X.
24. Lazon de la Jara P, Erickson D, Erickson P, Stapleton F. Visual and non-visual factors associated with patient satisfaction and quality of life in LASIK. Eye (Lond). 2011 Sep;25(9):1194-201. doi: 10.1038/eye.2011.151.
25. Llovet F, de Rojas V, Interlandi E, Martín C, Cobo-Soriano R, Ortega-Usobiaga J, Baviera J. Infectious keratitis in 204 586 LASIK procedures. Ophthalmology. 2010 Feb;117(2):232-8.e1-4. doi: 10.1016/j.ophtha.2009.07.011.
26.Stodola E. LASIK worldwide. EyeWorld.org. ACRS Apr 2016. Available at web.archive.org/web/20180612162458/https:/www.eyeworld.org/article-lasik-worldwide [accessed Nov 2025].
27. Stuart A. A look at LASIK past, present and future. 2009. EyeNet magazine. Archived from the original on 31 July 2013. Retrieved 12 September 2013. Available at: aao.org/eyenet/article/look-at-lasik-past-present-future [accessed Nov 2025].