mieducation

Managing Intraocular Pressure Rise:
Is There a Formula?
Two recent studies reviewing the long-term safety of soft contact lens (SCL) wear in children found low complication rates, similar to that of adults, with minimal impact on ocular physiology. 1,2This article summarises the key points within both papers and how they relate to fitting children with daily disposable contact lenses in clinical practice.
WRITER Dr Christolyn Raj
LEARNING OBJECTIVES
On completion of this CPD article participants should:
1. Understand the underlying mechanism causing intraocular pressure (IOP) rise in the acute or sub-acute setting, and the subsequent risk of secondary glaucoma,
2. Realise the importance of a patient’s history and treatment before formalising an effective management plan,
3. Be well-versed in treatment options for early, medium, and long-term stages of disease, and
4. Understand how to co-manage steroid-induced IOP fluctuations with other health care professionals.
Eye care professionals worldwide are managing patients who present with acute or sub-acute elevated intraocular pressure (IOP) in the context of past, present, or continued use of commonly prescribed corticosteroids.
Although this is a commonly presenting issue, there are challenges with managing this condition. These challenges predominantly relate to titrating the continued use of steroid medications with IOP-lowering agents, establishing a cautious management plan focused on achieving an acceptable IOP for the patient, and achieving our ultimate goal of preventing the sequela of secondary open-angle glaucoma.
In this article, Dr Christolyn Raj considers whether we can better predict patients who are likely to have an IOP spike after steroid use.
The increasing use of topical corticosteroid agents for eye conditions alone ranges from post-operative laser refractive surgery to cataract surgery, both routine and dropless, as well as glaucoma and retinal surgery. Some corticosteroid formulations have greater corneal penetrance, such as Prednefrin Forte (prednisolone acetate 1%, phenylephrine hydrochloride 0.12%) and increase the risk of IOP rise with prolonged use.1 In addition, corticosteroids employed in the management of ear, nose, and throat conditions from allergic rhinitis to sinusitis, as well as immunological, rheumatological, and connective tissue disease, often result in patients needing to remain on long-term steroids, impacting their inherent risk of developing secondary open-angle glaucoma.2,3
In this scenario, the golden adage of ‘do no harm’ as per the Hippocratic oath, plays a vital role where we, as clinicians, are faced with the difficult task of achieving a stable medical status while ensuring that vision is not being threatened.
DEFINING STEROID RESPONSE AND ASSOCIATED RISK FACTORS
Steroid-induced IOP rise was first described in the 1950s following the use of systemic adrenocorticotropic hormones and topical or systemic steroids.4 Since then, we have highlighted plausible risk factors associated with this phenomenon. Age, in particular, is a risk factor, with studies reporting that children and older adults have a more immediate and sustained IOP response to steroids.5 Steroid-related factors, including prolonged duration and potency of the steroid medication, have also been shown to be relevant. Other risk factors reported include previous spikes in IOP and a family history of glaucoma. Some associations that have been highlighted include myopia, a diagnosis of diabetes mellitus, connective tissue disease, or a history of uveitis.6,7 While it is still uncertain whether some patient characteristics – such as gender or race – play a role, some studies have suggested that males and African-Caribbean individuals are at greater risk.8
In some instances, investigating risk factors in individuals presenting with a steroid response can be elusive, however steroid response has been clinically defined. In the current literature, it is referred to as IOP above 21–24 mmHg and/or an increase of greater than five to 10 mmHg compared to baseline, often up to six weeks following use.9
Armalay and Becker10 reported a three-step graded classification of steroid response based on topical steroid use, which accounts for the majority of IOP rises.
The classification refers to:
1. Non-responders (66% of the population): IOP rise of less than 6 mmHg from baseline.
2. Moderate responders (33% of the population): IOP rise of 6–15 mmHg from baseline.
3. High responders (less than 6% of the population): IOP rise of more than 15 mmHg from baseline.
The above classification is a good guide for initial assessment and triage of patients with steroid response, however as always in glaucoma management, there are limitations to utilising IOP measurements alone to guide treatment. Measurement bias can cloud this evaluation and needs to be taken into account at each visit. In my practice, I use Goldmann tonometry as the gold-standard of measurement and use it whenever possible. I have found it is also useful to perform corneal pachymetry at baseline to rationalise IOP measurements, though this in itself can be affected in cases of anterior segment inflammation. And finally, IOP measurements alone are of little benefit without a thorough assessment of optic nerve function. This will ultimately guide our decision making with regard to the potency of IOP-lowering agents and duration of use. In my practice, a patient with a steroid response will enter into a glaucoma work-up assessment involving: gonioscopy, dilated fundus assessment of the disc using a 60D or similar retinal lens, ocular coherence tomography (OCT) imaging of the neuro-retinal rim (NRR) and retinal ganglion cell layer, as well as standard perimetry.
PUTTING OUR KNOWLEDGE INTO PRACTICE
Case A: A 65-year-old White male presented with a history of frequent use of multiple short courses of inhaled corticosteroids for chronic sinus disease over two years. Dennis Clark* had initially presented for routine review to his optometrist. He reported use of inhaled steroids six weeks ago. His visual acuity was 6/7 (Snellen) in each eye with a low hyperopic prescription. His IOPs were measured at 25 mmHg and 32 mmHg in the right and left eyes, respectively. Baseline IOPs were previously reported by his optometrist at 14 mmHg and 17 mmHg.
In consultation with Mr Clark’s physician, the inhaled steroids were replaced with a steroid-sparing agent, and initial treatment with a prostaglandin analogue was commenced.
Over the following two months, Mr Clark’s IOPs remained in the low to mid 20s. Further topical treatment with a combination agent was trialled, with little reduction in the IOP.
On clinical examination, gonioscopy revealed wide open angles, however the optic nerve appearance was suspicious for glaucomatous changes with loss of inferior and superior NRR most prominent in the right nerve (Figure 1). Interestingly, perimetry showed non-specific losses, which did not correlate with the optic nerve appearance (Figure 2). Given a higher than baseline IOP measurement despite medical treatment, and in the context of a glaucomatous disc appearance, a further reduction in IOP was determined to be necessary. Subsequent treatment with selective laser trabeculoplasty (SLT) was performed, achieving IOPs in the mid-teens. Mr Clark, who now has early-stage secondary open-angle glaucoma, continues to be managed with six-monthly reviews.
Case B: A 48-year-old White male presented on referral from his optometrist after reporting reduced vision over the past six months. Nick Smith* had a less clear history of steroid use, apart from describing being an atopic child who remained on inhaled steroid sprays for asthma and topical steroid creams for eczema into his teenage years. He described a strong family history of primary open-angle glaucoma (POAG), with his father having progressed to blindness from glaucoma in his early 60s.
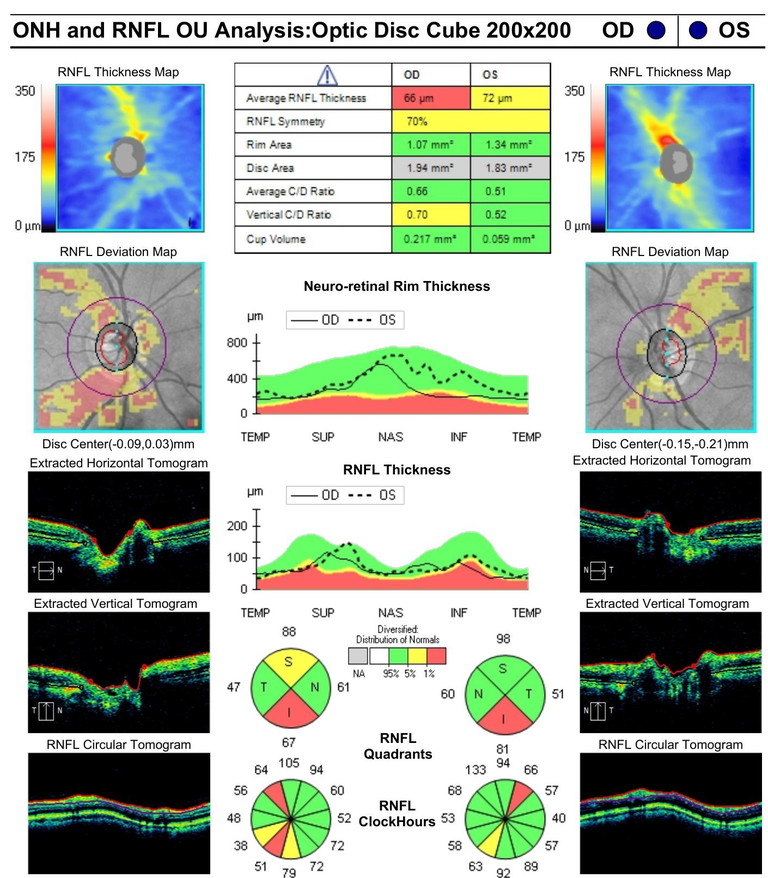
Figure 1. Case A: OCT showing optic nerve head appearance and neuro-retinal rim thinning, most prominent in the right eye.
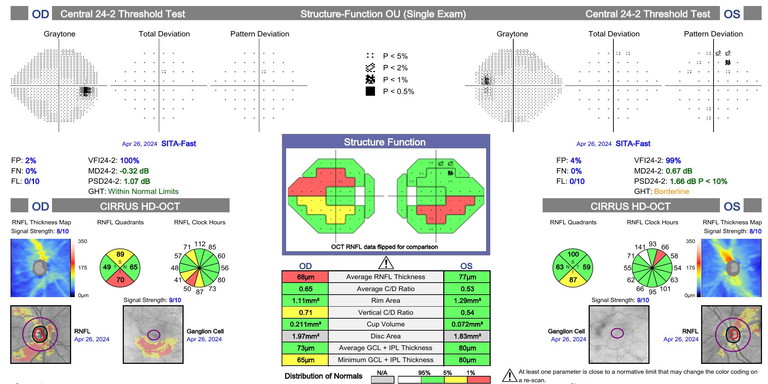
Figure 2. Case A: SITA-Fast perimetry results showing non-specific visual field losses.
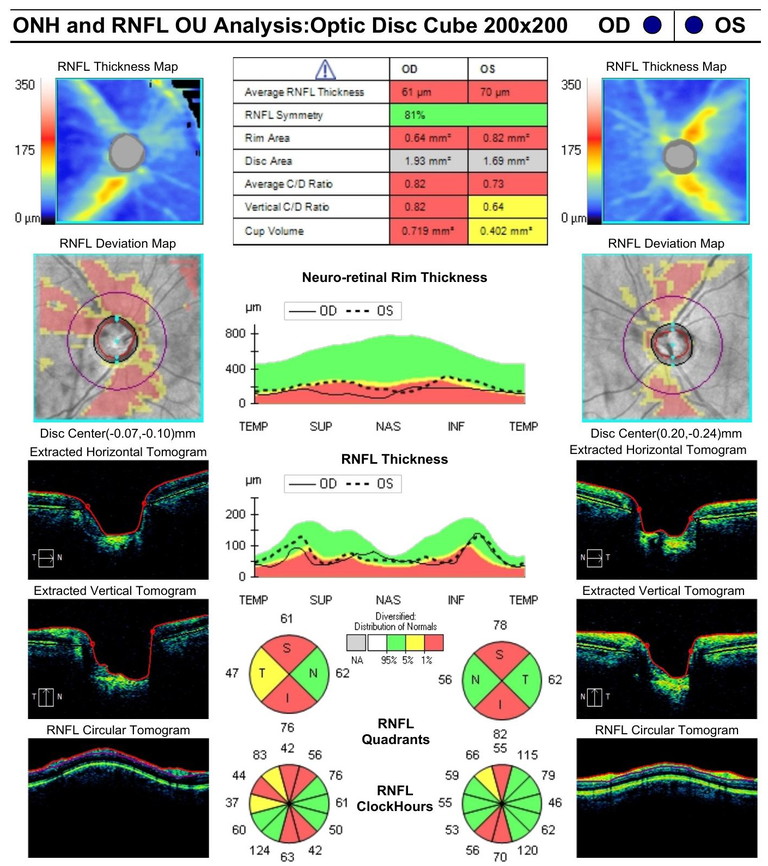
Figure 3. Case B: OCT showing advanced optic nerve head cupping in both eyes and neuro-retinal rim thinning.
On clinical examination, his best corrected visual acuity (BCVA) was 6/12 in the right eye and 6/9 in the left eye, with a myopic prescription (-3.50 DS). Mr Smith’s IOPs measured 27 mmHg and 23 mmHg in the right and left eyes, respectively. Unfortunately, there was no previous measurement to establish a clear baseline IOP.
Corneal pachymetry was 549 microns in the right eye and 554 microns in the left eye. Gonioscopy revealed open angles. Figure 3 shows advanced cupping in the optic nerve, with thin NRRs and cup-to-disc ratios in the order of 0.9 to 0.8 in the right and left eyes, respectively. Once again, perimetry showed non-specific losses, inconsistent with the nerve appearance. OCT showed significant macular retinal ganglion cell layer loss; most obvious in the right eye (Figure 4).
The cupped optic nerves, thin NRR, and loss of ganglion cells at the macula established a convincing clinical appearance of open-angle glaucoma. However, in the absence of perimetric findings, it did pose a management dilemma. Although it was plausible that Mr Smith’s myopic refractive error and thin corneas may have contributed to the disc cupping and elevated IOP measurements, his risk factors (previous heavy steroid use and strong family history of glaucoma) were extremely concerning for glaucoma progression.
Mr Smith was initially commenced on maximal topical therapy, which at six weeks showed some improvement with IOPs in the low 20s. Further management was postulated, including SLT and surgical intervention with minimally invasive glaucoma surgery (MIGS); however, the patient was subsequently lost to follow up.
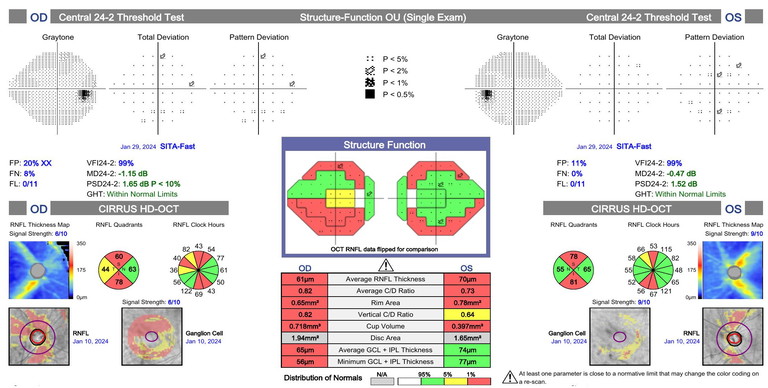
Figure 4. Case B: SITA-Fast perimetry results showing non-specific losses and macular retinal ganglion cell loss, most prominent in the right eye.
WHAT IS THE PATHOPHYSIOLOGY INVOLVED?
As the case studies highlight, presentation of steroid response can be insidious and there is not always a clear precedent of recent steroid use.
The underlying pathology of steroid-induced IOP rise is believed to be caused by increased resistance to the aqueous outflow at the level of the trabecular meshwork. It has been described that an increased production and decreased destruction of the extracellular matrix of the trabecular meshwork is likely responsible. The cells within the trabecular meshwork have glucocorticoid receptors, which steroids may target to alter cell migration and phagocytosis. In turn, this causes decreased cellularity of the meshwork and increased extracellular matrix deposition, thus increasing aqueous outflow resistance and a rise in IOP.11
It has been postulated that in some patients with connective tissue disease, such as rheumatoid arthritis, the ultrastructure of the trabecular meshwork may make them more susceptible to these steroid-induced changes. Similarly, in other patients with a strong family history of open-angle glaucoma, anatomical changes within the trabecular meshwork as a result of genetics may place them at greater risk. If left unattended, all these cases where aqueous outflow is impaired may result in irreversible vision loss.12,13
FORMULA… OR NOT?
This brings us to the fundamental question: can we better predict who is to have an IOP spike after steroid use? This is not unreasonable to consider, given that we have several parameters in medicine to determine individuals who are at risk of a disease process. A few examples include erythrocyte sedimentation rate (ESR) predicting the risk of giant cell arteritis (GCA), the cardiac calcium score predicting the risk of ischemic heart disease, and the Scoring Tool for Assessing Risk (STAR) calculator, which predicts the five-year risk of untreated ocular hypertension progressing to open-angle glaucoma.14-16
My research group is focused on this very question. To investigate steroid-related IOP response, we are analysing IOP measurements following routine cataract surgery with a standard topical steroid regimen, in a large cohort of patients of varying age, gender, and ethnicity.
While this retrospective analysis specifies strict exclusion criteria, such as patients with pre-existing POAG, uveitis, or inflammatory disease, it still has some limitations. It may not be possible to arrive at a generic formula based on baseline IOP and risk factors alone. Perhaps we only need to have a simple formula for ‘safe’ IOP range one month following steroid use. This could be enough to consider whether there has been a relative IOP rise, advise on how often we need to monitor such patients, and offer some guidance for follow up.
MANAGING THE ACUTE AND CHRONIC COURSE
When faced with patients who show a steroid response, what is the mantra for management? Unfortunately, there is no one-size-fits-all approach, however employing the following fundamental principles may help.
Always remember that the ultimate goal is to prevent progression to secondary open-angle glaucoma; lowering the IOP is simply the method by which this is done.
In my practice, I firstly ascertain whether the inciting agent, in this case steroids, can be ceased in the short or long term, or not at all. This will offer some guidance on whether you may be able to manage this patient under optometric care alone or may require co-management with an ophthalmologist and/or the patient’s physician. It is then necessary to establish the baseline IOP before defining an acceptable range of IOP following treatment.
I often find it easiest to address the ‘steroid use’ initially. If the patient is still on steroids, consider a tapering regimen depending on the presence of inflammation. For example, in cases of anterior uveitis, pseudophakic macular oedema, or scleritis, a slower taper over weeks may be required, whereas a fast taper may be sufficient in cases of corneal oedema. In some instances, where there is persistent inflammation requiring treatment over months, such as keratoconjunctivitis, a steroid-sparing agent, such as topical cyclosporin, may be needed.
Subsequent topical treatment with an IOP-lowering agent is then required, with viable options being prostaglandin analogues, beta blockers, and carbonic anhydrase inhibitors (CAIs). Again, we need to be prudent that these drugs do not interfere with a patient’s medical status; avoid beta blockers in cardiac and asthma patients, avoid CAIs in patients with a sulphur allergy, and prostaglandin analogues in patients prone to uveitis or maculopathy.
In some cases, adjunctive treatment in the form of laser or surgery may be required to achieve acceptable IOP readings in the context of progressive optic nerve damage. In this scenario, collaborative management with the patient’s physician is necessary to explore options for short-term cessation of steroids, use of a less potent steroid or steroid sparing agent. Most importantly, in all these cases, patients need to be followed closely with at least quarterly reviews, and with repeat imaging and regular perimetry to detect any sinister progression.
IS THIS THE SILENT THIEF OF SIGHT?
In hindsight, steroid-induced IOP rises may be the incipient cause of a secondary glaucoma in more patients than we expect. It has been postulated that elderly patients, who have received steroid treatment in the past, may present as normal-tension glaucoma.17 Other patients may not recall the use of steroids, even if it was infrequent, but should they pose some inherent risk factors such as a strong family history, connective tissue disease, etc., they may be more susceptible to even a short steroid course and need to be closely monitored.18
It is important, therefore, to ensure that we ask about steroid use – past and present – in every patient who presents as a ‘glaucoma suspect’. In the absence of a clear history, undertaking a brief general medical questionnaire may give a clue to the underlying aetiology. Patients with a history of allergic rhinitis, sinusitis or allergic conjunctivitis may have used topical or inhaled steroids in the past; those with a history of asthma or atopy may have been exposed to oral steroids when younger; all oncology patients, transplant recipients, patients with inflammatory bowel disease or connective tissue disease, are likely to similarly have had a course of steroids at some point in their treatment.
Eliciting this information will be crucial in developing a safe management plan for these patients, allowing us to triage their risk of progression, administer treatment with appropriate target IOPs, and escalate treatment if the latter is not met.
*Patient names changed for anonymity.
To earn your CPD hours from this activity, visit mieducation.com/managing-intraocular-pressure-rise-is-there-a-formula?

References available at mieducation.com.

Dr Christolyn Raj MBBS (Hons) MMED MPH FRANZCO is a cataract /refractive surgeon based in Melbourne. Dr Raj is a Fellow of the Royal Australian and New Zealand College of Ophthalmologists and the American Academy of Ophthalmology. She is a dedicated researcher through her affiliation with the University of Melbourne, Department of Medicine. Her research group is currently focused on improving patient outcomes from cataract surgery, with projects that include investigating post-operative intraocular rise and, in collaboration with the Royal Melbourne Institute of Technology University, development of a patient specific educational app on cataract surgery.