mieducation

The Intersection of Glaucoma and Cognitive Neurodegeneration
Glaucoma in the eye and cognitive neurodegeneration in the brain have many shared features from epidemiological, pathophysiological, and clinical perspectives. Yet, they remain intersecting but distinct, due to the breadth of heterogenous presentations of both in clinical practice.
This article provides an overview of the commonalities and clinical implications of glaucoma and cognitive neurodegeneration.
WRITERS Dr Jack Phu and Henrietta Wang
LEARNING OBJECTIVES
On completion of this CPD activity, participants should:
1. Understand the differences between dementia subtypes frequently seen in clinical practice,
2. Understand the shared pathophysiological mechanisms between dementia and glaucoma, and
3. Understand the similarities and differences in structural and functional measurements when comparing dementia and glaucoma.
The eye is an extension of the brain, with the optic nerve representing the second cranial nerve of the central nervous system.1 At its simplest, the sensation of vision is first triggered by phototransduction as contrast: differences in luminance and visual properties between objects and backgrounds.2,3 Then, signals from the eye are sent to the brain via the optic nerve, and undergo further processing in the cortices, providing meaning to the simple visual information.4,5 Therefore, the process of vision encompasses reception of signal and its interpretation, before decisions are made in higher executive areas.
Consequently, age-related or pathological changes occurring in one part of the central nervous system may manifest with signs in the other.6 The changes are crucial to an individual’s daily function, as among the senses, vision represents the major modalities for interacting with the external environment. Given this, there is a desire to understand the links between the eye and brain in terms of age- and pathology-related changes.
A specific topic of interest is the intersection of glaucoma and cognitive neurodegeneration, specifically Alzheimer’s disease and other forms of dementia.
DEMENTIA AND COGNITIVE NEURODEGENERATION
Dementia describes a clinical endpoint, representing cognitive changes as a common feature of many neurodegenerative aetiologies.7 Typical symptoms of dementia include a decline in memory, thinking, behaviour, motor control, emotion, and language functions; the relative extent to which these domains are affected may also relate to the underlying diagnosis.8
The most common cause of dementia worldwide is Alzheimer’s disease, followed by vascular dementia, Lewy body dementia, and frontotemporal dementia (Table 1).8 Each phenotype of dementia has its own diagnostic criteria.9-11 Currently, most criteria are clinical, with a significant emphasis on the functional impact.8 Thus, diagnoses of these conditions involve a core battery of neuropsychology tests, supplemented by neuroimaging and other special tests.
Risk factors for dementia relate to the underlying pathology. In general, age is the strongest risk factor for dementia and cognitive neurodegeneration, with the risk approximately doubling every five years after age 65 years.12 Despite frontotemporal dementia affecting a primarily younger cohort of people, age as a risk factor applies to all major causes of dementia.
Specifically relevant to Alzheimer’s disease, the most common cause of dementia, genetic factors (APOE (Apolipoprotein E) gene e4 allele or other autosomal dominant mutations including APP, PSEN1 and PSEN2) play a particularly important role in risk.13,14 However, this is not routinely examined in clinical practice except in cases of suspicious family history, especially in early-onset Alzheimer’s disease.
Recently, the Lancet Commission highlighted an important statistic: approximately 40–45% of dementia cases worldwide may be attributable to potentially modifiable risk factors.7 The list of modifiable risk factors is categorised into the following: 1) cardiovascular and metabolic (especially midlife hypertension); 2) education and cognitive reserve; 3) hearing and vision impairment; 4) lifestyle factors; 5) mental health (especially depression); 6) social factors; 7) traumatic brain injury; and 8) environmental and other medical factors.
Notably relevant to this discussion, untreated vision impairment is a recognised risk factor.15-18 Holistically, the mechanism underpinning sensory impairment as a modifiable risk factor for dementia involves the epiphenomena of social isolation, increased cognitive load, and reduced sensory input. While hearing loss is one of the strongest modifiable risk factors, the contribution of vision impairment is something that can be addressed by eye care clinicians.
An obvious cause of reversible vision impairment is uncorrected refractive error, which is relatively simple to correct. Although the blindness resulting from glaucoma is irreversible, it is possible in many patients to treat and manage the disease appropriately, to significantly reduce the risk of visual impairment.
Therefore, correction of reversible causes of visual impairment, or efforts to prevent irreversible vision loss, are important considerations for preventing morbidity and mortality associated with dementia and cognitive neurodegeneration.7
In the next section, the epidemiological commonalities between dementia and glaucoma are examined.
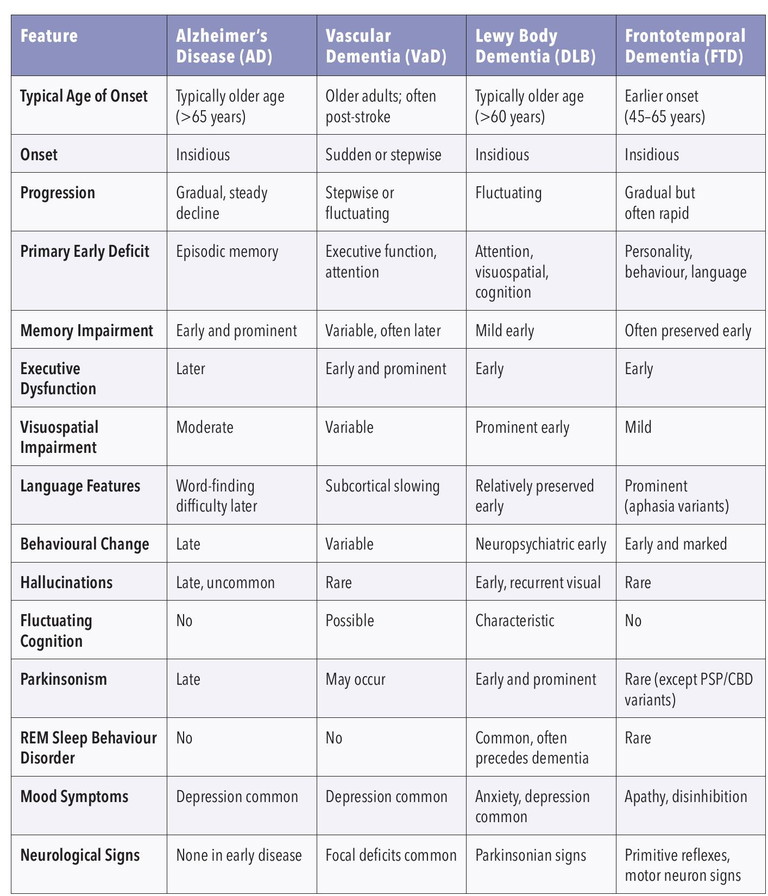
Table 1. Differences and similarities in the clinical features of the top four causes of dementia.
EPIDEMIOLOGICAL COMMONALITIES
Worldwide, there are over 55 million people living with dementia, with an increase in prevalence yearly due to an ageing population.19 Although the relative prevalence and incidence rates have remained relatively stable over time, the absolute case numbers have increased sharply due to population growth and ageing. Most dementia occurs in older adults (aged 65 years and over), and women are disproportionately affected.12
Dementia affects over 420,000 Australians.20 It is a leading cause of death in the older population. In the next 40 years, this number is expected to double.
In comparison, glaucoma affects over 75 million people worldwide,21 and over 300,000 people in Australia.22 Similarly, there is an increased prevalence with age, but with a lower limit of age 40 years as a significant cut-off for the increased risk.
Commonalities in the epidemiology of dementia and glaucoma reflect mechanistic pathways involving age-related vulnerabilities and degeneration. Accordingly, epidemiological studies have been conducted to seek links between the two conditions.
A recent meta-analysis, including over nine million individuals, found that glaucoma was associated with an increased risk of all-cause dementia and its specific aetiologies.23 As expected, there was a higher prevalence of both conditions, particularly in older adults. Specifically, the authors suggested that primary open-angle glaucoma, in comparison to other types of glaucoma, was more likely to lead to dementia. Interestingly, the authors reported that glaucoma was not associated with dementia in Europe and North America, but was associated in other regions.
A national cohort study involving 16 million person-years of follow-up found that individuals with glaucoma had a 1.39 times risk of Alzheimer’s disease, 1.66 times risk of vascular dementia, and 1.57 times risk of all-cause dementia.24 Again, older age was associated with greater risk.
However, other studies have highlighted potential confounders of the relationships. A large meta-analysis of cohort studies involving over 4.6 million individuals showed no increased risk of incident Alzheimer’s disease in people with glaucoma (1.03 times risk, with a confidence interval of 0.93–1.05).25 Similar results were found for other causes of dementia. This study argued that not accounting for confounding factors artificially strengthens the relationship between glaucoma and dementia. Instead, accounting for factors such as age and genetics leads to glaucoma not being an independent risk factor for dementia.
Overall, although there are common pathophysiological pathways between glaucoma and dementia, it is unlikely that one directly affects the other. Instead, it is important to adjust for common risk factors such as age, vascular disease and, in the case of dementia, cognitive baseline when interpreting epidemiological risk.25 The next section will examine the commonalities in the pathophysiological pathway.
SHARED PATHOPHYSIOLOGICAL PATHWAYS
Both glaucoma26 and Alzheimer’s disease27 (as the leading cause of dementia) are represented by a progressive loss of neurons, chiefly in the retinal ganglion cells and cortical and hippocampal regions, respectively. In glaucoma, the locus of change is the retinal ganglion cells. In Alzheimer’s disease, degenerative changes predominantly occur at the cortex and hippocampal regions.
Features of glaucoma, such as the subset of patients in whom intraocular pressures are purportedly ‘normal’ or ‘well-controlled’ yet still demonstrate progressive structural and functional loss, suggest additional pathophysiological factors at play.28
The links between glaucoma and cognitive neurodegeneration may be divided into several areas: their shared molecular pathology, neuroinflammation and glial dysfunction, mitochondrial dysfunction and oxidative stress, and neurovascular dysfunction. These are summarised in Figure 1. Overall, this culminates in axonal transport failure and manifests as transsynaptic degeneration – retrograde from brain to eye and anterograde from eye to brain, as seen in Figure 2.
SHARED MOLECULAR PATHOLOGY
Several studies have demonstrated shared molecular pathology between the eye and brain. Specifically, these include amyloid-beta (A-beta) accumulation and tau pathology.
Amyloid-beta has been identified in retinal tissue, the optic nerve, and the lateral geniculate nucleus in experimental glaucoma models in primates.29,30 It has also been found in the retina in post-mortem studies of people with Alzheimer’s disease.31 Some animal models have shown amyloid-beta deposition preceding the onset of retinal ganglion cell loss, suggesting a potential causative pathway, rather than an epiphenomenon.32 The pathophysiology of amyloid-beta in neurodegenerative disease is its induction of synaptic dysfunction, mitochondrial toxicity, oxidative stress, and microglial activation.
Tau has been suggested to have stronger links to glaucoma. It is normally a microtubule-associated protein responsible for axonal stability, axonal transport, and neuronal polarity. In pathology, tau becomes hyperphosphorylated, detaches from microtubes, and aggregates into neurofibrillary tangles, which disrupt axonal transport. Hyperphosphorylated tau has also been detected in retinal ganglion cells, optic nerve axons, and the visual cortex in models of glaucoma.33,34 In Alzheimer’s disease, tau in the retina may serve as a biomarker and may precede observed brain pathology.35
NEUROINFLAMMATION AND GLIAL CELL DYSFUNCTION
Two key glial cells in the central nervous system are astrocytes and microglia.36,37 Both provide distinct roles in supporting the nervous system.
Microglial cells are the immune cells within the brain. Their roles include acting as first responders to injury, infection and disease, brain development, and synaptic pruning. Within their role as the primary immune cells within the brain, specific functions of glial cells include immune surveillance, phagocytosis, neuroinflammation, and tissue repair.
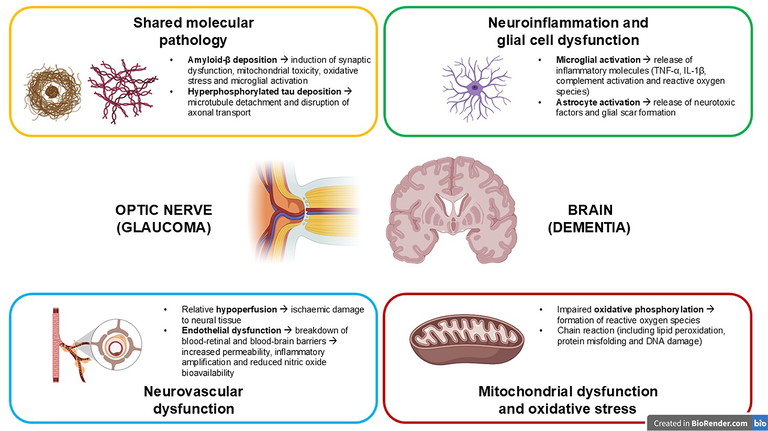
Figure 1. A summary of several pathophysiological pathways shared between degenerative eye and brain pathologies.
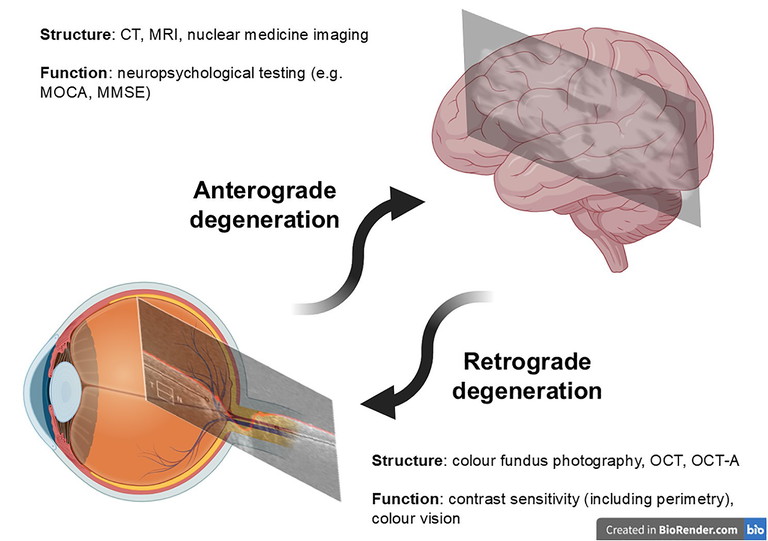
Figure 2. Anterograde and retrograde degeneration occurring between primary eye and brain pathologies, and examples of structural and functional measurements of neural integrity.
In both glaucoma and Alzheimer’s disease, there is activation of microglial cells, which has been observed at the optic nerve and retina (in glaucoma),38,39 and in the cortex and hippocampus (in Alzheimer’s disease).40,41 The chronic pathological activation of microglial cells leads to a shift from protective to pro-inflammatory phenotypes, with release of inflammatory molecules such as TNF-α, IL-1β complement activation, and reactive oxygen species. Accordingly, these lead to cellular damage.
Astrocytes play important roles in structural support (including maintaining the blood-brain barrier) and homeostasis, metabolic support, synaptic regulation, and neuroprotection (including glutamate clearance). In response to injury or insult, astrocytes become hypertrophic and can release neurotoxic factors, contributing to glial scar formation. Thus, dysfunction of astrocytes can lead to an exaggerated immune response and excitotoxicity. These processes have been demonstrated in both glaucoma and dementia.42-44
MITOCHONDRIAL DYSFUNCTION AND OXIDATIVE STRESS
Retinal ganglion cells and cortical neurons are energy-intensive. As the powerhouse of the cell, damage to mitochondria leads to impaired oxidative phosphorylation (efficient energy production) and, therefore, the formation of reactive oxygen species.45 Reduced mitochondrial function has been demonstrated in the retinal ganglion cells in glaucoma46,47 and in cortical neurons in Alzheimer’s disease.48
Impairments in mitochondrial function and the activated microglia converge to oxidative stress. An additional factor of clinical relevance is impaired antioxidant defences, which normally reduce the burden of reactive oxygen species. This may potentially be targeted by novel therapeutic agents. Oxidative stress represents a family of processes that consequently lead to a chain of reactions, including lipid peroxidation, protein misfolding, and DNA damage.49
NEUROVASCULAR DYSFUNCTION
Energy delivery to the neural tissue is contingent on effective blood flow. There is a tight window of ideal flow and pressure characteristics, with extremes of high or low being associated with stress and damage.
Both glaucoma50 and dementia51 have demonstrably reduced blood flow, which has been associated with impairments in autoregulatory mechanisms. Autoregulation is the mechanism by which stable perfusion is maintained under stress conditions. In conjunction with individually increased susceptibility to hypoperfusion-related stress, this eventually leads to ischaemic damage to neural tissue.
Another manifestation of neurovascular dysfunction is endothelial dysfunction, which is normally important for maintaining the blood-retinal (eye) and blood-brain barriers. Dysfunction leads to increased permeability, inflammatory amplification, and reduced nitric oxide bioavailability.
As described above, the vascular pathway to glaucoma has long been recognised as an alternative explanation for why people with intraocular pressures within the ‘normative range’ may still develop glaucoma or progression. Similarly, hypoperfusion in the brain, such as in recognised risk factors including hypertension, is a recognised contributor to Alzheimer’s disease pathology.52
GENETIC AND MOLECULAR OVERLAP
The pathophysiological pathways described above are not exclusive to diseases such as glaucoma or cognitive neurodegeneration. For example, reactive oxygen species play important roles in numerous diseases, especially those related to age-related deterioration, including age-related macular degeneration.53 The question is whether these pathways may be particularly relevant in people who are at increased risk for both glaucoma and cognitive neurodegeneration.
Recent genome-wide analysis studies have demonstrated pleiotropy between glaucoma and Alzheimer’s disease.54 Pleiotropy is defined as the phenomenon in which a single gene influences multiple phenotypic traits. This occurs since gene products, such as proteins, may be used in different tissues at different stages of development, ultimately leading to a variety of effects.
Taking the shared pathophysiological pathways into context, pleiotropy could explain mutual defects in processes, such as mitochondrial metabolism, immune regulation, and cell survival/apoptosis.
Since there are genetic factors that appear to be exclusively relevant to glaucoma and Alzheimer’s disease, the concept of genetic and molecular overlap refers to the neuronal vulnerability arising from certain genetic variants. These variants do not cause the diseases, but modulate individual susceptibility, which in turn is affected by other contextual factors described above.
To highlight the exclusivity, we could consider the example of APOE e4, the gene that is strongly pleiotropic for Alzheimer’s disease and vascular dementia.13 However, it has not been demonstrated to be strongly pleiotropic for glaucoma.55 Therefore, while amyloid may appear or co-exist in glaucoma, glaucoma-related pleiotropy lies downstream of amyloid biology.
RETINAL INSIGHTS INTO THE BRAIN
The eye has been investigated as a proxy for detecting neurodegenerative changes occurring in the brain. Similar to how glaucoma diagnosis is contingent upon structural and functional changes, retinal insights into brain pathology can also be considered in the following broad categories of the ophthalmic examination:
Retinal Structural Changes in Dementia
The proliferation of optical coherence tomography (OCT) imaging has provided micron-level resolution of retinal – and therefore neural – tissue. Multiple OCT studies have demonstrated retinal nerve fibre layer (RNFL) thinning in Alzheimer’s disease and mild cognitive impairment.56 The thinning occurs most commonly in the superior and inferior quadrants.56 Another study has also found RNFL thinning associated with poorer scores on neurocognitive testing.57
In comparison to the characteristic nerve fibre bundle defects occurring in glaucoma, such as in arcuate patterns, the defects seen in dementia are non-specific.58 The defects are also not associated with changes to the optic cup. Nonetheless, the RNFL defects in cognitive neurodegeneration can confound structural analysis in coexistent or suspected glaucoma.
The ganglion cell layer (GCL or in combination with the inner plexiform layer, GCIPL) can also be imaged using OCT. In theory, GCIPL may be a more specific marker of neuronal cell body loss, rather than its downstream axon. Studies have shown similar GCIPL thinning occurring in the context of cortical thinning and cognitive impairment.59 Again, the loss tends to be diffuse compared to the sectoral patterns in glaucoma.60
Macular thickness in its entirety is rarely used in the glaucoma assessment as it encompasses tissue layers that are, at least initially, not affected in glaucoma. Interestingly, macular thickness measurements have also been correlated with neurocognitive testing in the context of dementia.61
“People with cognitive impairment may present a challenge in the clinical examination”
From an experimental perspective, amyloid-beta and tau – markers of dementia pathology – have been detected in retinas in post-mortem histopathology studies.62 As mentioned previously, these have been seen in the retinal ganglion cell and inner plexiform layers. However, imaging of these pathological features is not yet ready for clinical practice.
More recently, OCT angiography (OCT-A) has become more widely available in clinical practice. Given the associations between vascular irregularities and the incidence of glaucoma and dementia, OCT-A may provide complementary information to structural OCT. OCT-A may also serve as a surrogate marker of amyloid plaque burden in the brain. Several studies have shown generalised features of hypoperfusion: reduced vessel density, enlargement of the foveal avascular zone, and capillary dropout.63 Again, there is overlap with features of glaucoma, except that the losses tend to be more diffuse, rather than focal, in dementia.64
Visual Changes in Dementia
Functional correlates are equally important in both glaucoma and dementia. Specific domains of interest include contrast sensitivity, colour vision, visuospatial skills, and higher-order visual dysfunction.
Some of these deficits in vision, such as the visuospatial domain, are part of diagnostic criteria for various aetiologies of dementia.
Contrast sensitivity is the ability of the visual system to resolve differences between a target and its background. Reductions in contrast sensitivity have been demonstrated in dementia, even in the context of normal visual acuity and an otherwise normal structural examination.65,66 Mechanistically, it is thought to be related to damage to cortical areas. Functionally, the impact on patients is difficulty with tasks such as driving (especially at dusk and night), reading low contrast text, and navigating uneven surfaces.
Perimetry assesses contrast sensitivity across the visual field. It has been well-documented that key perimetric parameters, such as mean deviation and pattern standard deviation, are worse in dementia compared to age-matched controls.67 Interestingly, differences have recently been highlighted in traditional reliability indices (fixation losses, false positives, and false negatives) compared to age-matched controls.67 This feature also highlights the cortical impairment in dementia, as these catch trials, in part, aim to capture the patient’s ability to maintain a cognitive load throughout the test. Some studies have highlighted relationships between cognitive scores and reliability indices.
An older study revealed colour vision abnormalities in dementia. In early stages, blue-yellow (tritan) deficits predominate, with relative preservation of the red-green axis – in a pattern similar to that of glaucoma.68 Like in contrast sensitivity, it is thought to reflect cortical, rather than retinal, changes.
Contrast sensitivity and colour vision are both clinical measures in the ophthalmic setting (such as using a Mars Contrast Sensitivity (MARS) chart or D-15 colour vision test, respectively). From a neuropsychological perspective, visual skills, such as visuospatial and visuoperceptual domains, are likely to reflect more upstream processes in the cortex and are therefore of interest in the diagnosis of dementia. These domains are assessed, even in basic clinical tests of cognition, such as the Montreal Cognitive Assessment (MoCA) and the Mini-Mental State Examination (MMSE).69,70
Visuospatial dysfunction may manifest with difficulties in depth perception, spatial orientation or neglect.71 Mechanistically, this is due to parietal lobe involvement, with disruption to the integration of visual, vestibular and proprioceptive inputs. Functionally, patients may be more prone to misjudging steps, kerbs and object locations, and may outwardly appear clumsy or hesitant.
Neglect is a non-specific sign that may occur in regions of profound vision loss. Classic examples of this, from an ophthalmic perspective, are a hemianopia or advanced glaucoma. Overtly, this may present with difficulties attending to complex scenes, visual crowding, and the appearance of inattention. Key regions of the brain involved in neglect are the inferior parietal lobule, the temporoparietal junction, the frontal eye fields, and the cingulate cortex.72 In combination, these structures govern the division and allocation of attention.73 Notably, neglect is particularly pronounced in right hemispheric damage (left-sided neglect).
Similarly, visual inattention and challenges with crowding are other domains affected. These manifest with difficulties in shifting gaze, sustaining visual attention, and filtering irrelevant stimuli.74
Visuoperceptual abnormalities can be broadly considered as visual agnosia or face/emotion recognition. Visual agnosia is the failure to recognise objects despite having intact vision (i.e. primary visual functions are preserved). This is due to damage to the ventral visual pathway (inferior temporal cortex).75
Though not a specific visual change, hallucinations may also be present in dementia. This is a classic hallmark feature of Lewy body dementia, but may be mistaken for Charles Bonnet syndrome, which may occur in advanced vision loss.
PRACTICAL ISSUES IN THE CLINIC
People with cognitive impairment may present a challenge in the clinical examination.
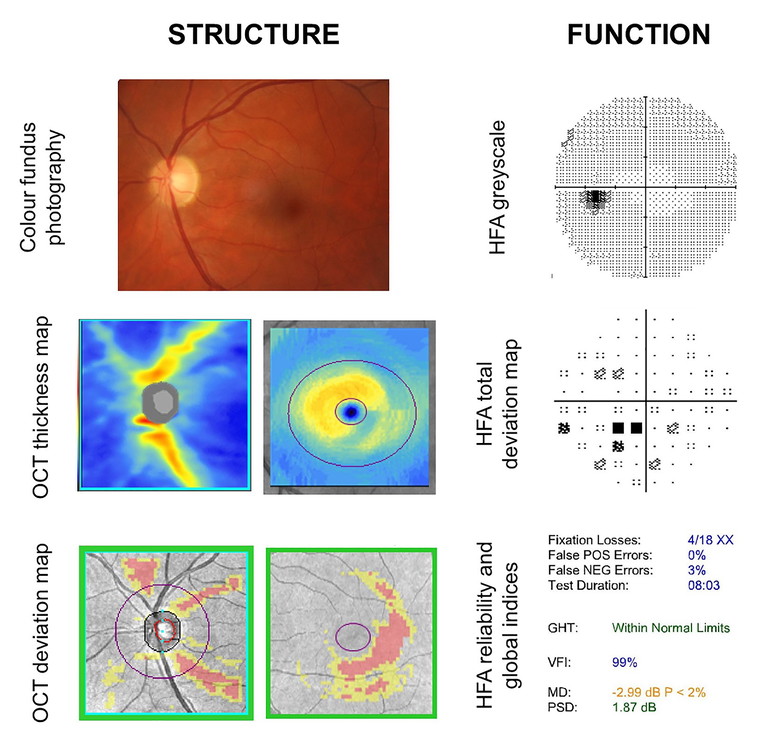
Figure 3. A case of a 52-year-old patient with early-onset dementia, seen to assess for glaucoma. Colour fundus photography revealed a slightly small disc with small cup and intact neuroretinal rim, but with non-contiguous retinal nerve fibre layer defects. OCT imaging confirmed the presence of structural defects. The functional defects assessed using the Humphrey Field Analyser (30-2 test grid, SITA-Standard) showed generalised loss on the total deviation map. Traditional reliability indices showed some catch trial errors, and the mean deviation result was statistically significantly reduced at -2.99 dB.
Relevant to glaucoma, the impact of cognitive impairment on the clinical flow includes clinical attendance, the reliability of testing, and additionally medication adherence. Each of these issues can lead to poorer clinical outcomes for patients.
Currently, structural and functional testing in clinical settings do not return specific signs for dementia. However, a holistic interpretation of the clinical examination may provide useful insights into cognitive ability, which could then trigger an appropriate diagnostic pathway for the patient.
Although glaucoma and dementia represent neurodegenerative processes in the central nervous system and share many pathophysiological features, they remain distinct clinical entities. An example of a 52-year-old patient with early-onset dementia referred for glaucoma assessment is shown in Figure 3. Here, there are marked structural defects seen on clinical examination and ocular imaging. However, these defects are not contiguous to the disc. While it is possible for glaucoma to affect both hemifields simultaneously, it is not likely in the context of a mild generalised visual field deficit. The marked structure-function discordance also raises the concern of a non-glaucomatous aetiology. There are commonalities in clinical features, but no conclusive evidence of causality.
One of the biggest clinical challenges remains the separation between the clinical presentations. Importantly, glaucoma is a diagnosis of exclusion, and causes such as cognitive neurodegeneration should be ruled out before committing a person to a pathway of glaucoma management.
To earn your CPD hours from this article, scan the code or visit mieducation.com/the-intersec-tion-of-glaucoma-andcognitive-neurodegeneration.

References available at mieducation.com.

Henrietta Wang BOptom (Hons) BSc MPH FAAO is an optometry-trained clinician-researcher. Her clinical, teaching and research duties are focused on ocular diseases, imaging, and collaborative care models. She has served on numerous national and international professional committees to advance the profession.

Dr Jack Phu MD PhD BOptom (Hons) Diplomate (Glaucoma) is a clinician scientist, honorary lecturer at University of New South Wales Sydney, Adjunct Professor at University of Houston College of Optometry, and junior medical officer at Bankstown-Lidcombe Hospital. His basic and clinical sciences research program focuses on improving the understanding of glaucoma and retinal disease, and its diagnosis and management in clinical practice.