minews

AREDS2 Nutritional Supplements for AMD: Where are we in Australia in 2026?
Age-related macular degeneration (AMD) is one of the leading causes of blindness and visual impairment in Australia and the Western world.1-4The Australian National Eye Health Survey conducted in 2016 found that 0.96% of non-indigenous Australians over 50 years of age have late-stage AMD, and 10.5% have intermediate-stage AMD.1 More recently, the Australian Eye and Ear Health Survey (AEEHS), released in July 2025, found that AMD was the leading cause of bilateral blindness in Australia, and one of the leading causes of both unilateral and bilateral visual impairment.2 The prevalence of AMD increases significantly with age, and this is of particular concern given the ageing population in Australia.1 It is currently estimated that 1.9 million Australians have some form of AMD, and this is set to increase to 2.1 million by 2030.5 On a global scale, the prevalence of AMD is estimated to increase to 288 million by 2040.6
WRITER Inez Hsing
LEARNING OBJECTIVES
On completion of this CPD activity, participants should:
1. Be aware of the prevalence of age-related macular degeneration in Australia and current treatments available,
2. Be aware of the role antioxidant supplements play in slowing progression of AMD and GA,
3. Realise the mechanism of action of AREDS and AREDS2 formulas, and
4. Realise optometrists’ role in educating and recommending supplements to AMD patients.
AMD is an acquired condition in which the retinal pigment epithelium (RPE) and overlying photoreceptors progressively degenerate due to a complex interplay of age, genetics, and exposure to both endogenous and environmental oxidative stress.7 Family history is known to be a risk factor for AMD – there are 52 common AMD-associated gene variants that can influence the immune system (complement pathway) and inflammatory responses thought to be associated with AMD, such as the CFH and ARMS2/HTRA1 gene variants.7,8 Exogenous oxidative stress, such as smoking, can contribute further to RPE insult and decompensation,7,9,10 and a sedentary lifestyle without sufficient physical activity likely increases overall inflammation within the body.11 Natural progression of AMD to late disease results in death of the overlying RPE and photoreceptors, characterised by centre-involving geographic atrophy (GA), or development of abnormal angiogenic vessels (neovascular AMD).7,10,12 The likelihood of progression from early-to-intermediate AMD to late AMD has been described in the literature from 15–20%13 to as high as 47%.14,15
At the time of writing, several intravitreal anti-VEGF agents are funded by the Pharmaceutical Benefits Scheme (PBS) in Australia for treatment of neovascular AMD, including aflibercept, ranibizumab, faricimab, and brolucizumab (for refractory cases). On the other hand, there are limited treatment options for GA – two intravitreal complement inhibitors, pegcetacoplan (Syfovre, Apellis) and avacincaptad pegol (Izervay, Astellas), were approved by the Therapeutic Goods Administration (TGA) for treatment of GA in 2025. These complement inhibitors have shown reduction in GA growth in Phase 3 clinical trials,16 however the AREDS2 Research Group recently reported that “limitations of both drugs include administration by repeated intravitreal injection every one to two months, relatively modest efficacy, important side effects (including substantially increased risk of new neovascular AMD, committing an eye to potentially lifelong anti-VEGF injections), and high cost (>US$2000 per injection)”.17 Thus, it is still important for Australian optometrists to understand and remain up-to-date on the benefits of AREDS2 nutritional supplements alongside appropriate diet and lifestyle changes in slowing AMD progression.
Presented here is an updated review of AREDS2 nutritional supplements in slowing the progression of intermediate AMD to late AMD. This article also highlights the important role that Australian optometrists play in educating patients with AMD, with particular emphasis on when to recommend AREDS2 antioxidant supplements and the importance of encouraging adherence.
DEVELOPMENT OF THE AREDS2 FORMULATION
The original AREDS formula was developed in 2001 following a large, randomised, multi-centre, double-masked, and placebo-controlled clinical trial of 4,757 patients over a mean follow-up period of 6.3 years.18 The formula consisted of antioxidants (500 mg vitamin C, 400 IU vitamin E, and 15 mg beta-carotene), 80 mg zinc (as zinc oxide) and 2 mg copper (as cupric oxide). Both vitamin C and vitamin E play an important role in minimising oxidative damage in the eye.11 Beta-carotene is a carotenoid agent not normally found in the eye,19 however it does share a similar molecular structure to xanthophyll agents (lutein and zeaxanthin) within the macula and was therefore thought to have fairly similar properties.20 Additionally, at the time of the AREDS study, only beta-carotene was readily available.18
Zinc is an essential trace element that is involved in numerous physiological processes within the body, including modulation of metabolic and oxidative reactions.21,22 It is found in high concentrations in the photoreceptors and RPE.11 The inclusion of copper was primarily to prevent neutropenia and anaemia caused by copper deficiency, which is a potential adverse effect of high-dose zinc.18,23 The severity of AMD was categorised according to Table 1 and this was used by the AREDS group to standardise grading of AMD in practice, and to more accurately assess disease progression.
The results of the AREDS study were very promising – in patients who had AREDS category 3 (intermediate) AMD, or where one eye had AREDS category 4 (late/advanced) AMD, the risk of category 3 eyes progressing to AREDS category 4 was reduced by 25% over five years compared to placebo.18 There was also a corresponding 27% reduction in the risk of losing greater than 15 letters on a standard ETDRS (Early Treatment Diabetic Retinopathy Study) logMAR chart.18 The risk of progression to advanced AMD was low in AREDS category 1 and AREDS category 2 (1% and 1.3%, respectively, over five years); therefore, it was not possible to draw any meaningful conclusions as to whether the use of AREDS antioxidants was indicated in this cohort.18 A follow-up observation study showed that the protective effect of AREDS antioxidant supplements persisted out to 10 years – those patients who had initially received the AREDS formula continued to demonstrate lower rates of progression to advanced AMD (particularly choroidal neovascularisation) and lower rates of both moderate and severe vision loss.24
Table 1. The Age-Related Eye Disease Study (AREDS) grading system for AMD (adapted from Al-Zamil and Yassin).7
There was, however, concern regarding the use of beta-carotene in current and past smokers; studies have shown that daily supplementation with beta-carotene in this group of subjects resulted in higher rates of lung cancer (two-fold risk)25 and associated mortality.26 Some of these studies were published during the course of the AREDS study. Subsequently, the 8% of the AREDS group who were smokers at the time of initial enrolment were offered the chance to withdraw from the study or to be re-assigned to a treatment arm not containing beta-carotene. This may have influenced why the rate of lung cancer in the AREDS cohort was overall low (0.8% over five years)18 but nonetheless, it raised the question whether alternate carotenoid agents with a better safety profile could be used.
Additionally, the recommended dosage of 80 mg zinc oxide in the AREDS formula exceeded both the tolerable upper intake level (UL) of 40 mg/day established by the United States Institute of Medicine27 and the TGA maximum recommended daily dose of 50 mg.28,29 Increased consumption of zinc has been shown to be affiliated with nausea and gastrointestinal upset, such as vomiting, epigastric pain, abdominal cramps, and diarrhoea.20,22 Long-term high-dose zinc can even potentially increase the risk of prostate cancer,30 copper-deficiency (which in turn can result in anaemia and neutropenia),23,31 and result in dysregulation of zinc homeostasis in the brain that could contribute to the development and progression of Alzheimer’s disease.22
To address concerns regarding beta-carotene and high-dose zinc, the AREDS2 Research Group followed 4,203 patients as part of a multi-centre, randomised, double-masked clinical and placebo-controlled trial conducted over five years.32 By this stage, lutein and zeaxanthin, both xanthophyll carotenoids found naturally concentrated within the macula, had become commercially available. Collectively, known as ‘macular pigments’, xanthophyll carotenoids give the macula its characteristic yellow appearance.20 Lutein and zeaxanthin have both antioxidative and anti-inflammatory properties and are responsible for filtering out short-wavelength blue light before it can reach the photoreceptors.19,33 Animal studies have also shown that these macular pigments may help regulate normal function of the blood vessels within the retina and choroid.34
Given its pivotal role in the macula, it had been postulated that increased intake of lutein in both healthy eyes and eyes affected by AMD could improve visual function and protect against the development of late AMD.35,36 Humans are incapable of synthesising lutein and zeaxanthin and instead rely on dietary or supplementary intake to obtain required levels.11 A meta-analysis conducted by Ma and colleagues33 found that increased dietary intake of lutein and zeaxanthin may be protective against development of late AMD but not early AMD. Similarly, Huang et al.37 established that supplementation with lutein and zeaxanthin increased macular pigment density within the macula and also increased central retinal sensitivity, as measured by both multifocal electroretinogram and microperimetry.37
Additionally, there was growing interest in the potential benefits of omega 3 long-chain polyunsaturated fatty acids (LCPUFA) on reducing the risk of progression to late AMD.11,38-41 Docosahexaenoic acid (DHA) is a major structural lipid found within the retina, and eicosapentaenoic acid (EPA) is the precursor to DHA. These LCPUFAs are found primarily within the photoreceptor outer segments and play a role in modulating the permeability of photoreceptor membranes.11,42 In the diet, they can be found in high concentrations in oily fish.11,41 It has been hypothesised that both DHA and EPA have anti-inflammatory and anti-atherosclerotic properties for the body, including the brain, cardiovascular system, inflammatory arthritic disease, and the retina.41,43 Sneddon and colleagues in 200140 were among the first to describe a possible protective effect of omega 3 and fish intake against AMD progression.
Thus, the primary aim of the ARED2 study was to determine whether the addition of lutein and zeaxanthin and/or DHA and EPA to the original AREDS formula would help to further reduce the risk of progression to late AMD.24 Patients were randomly assigned to one of four groups:
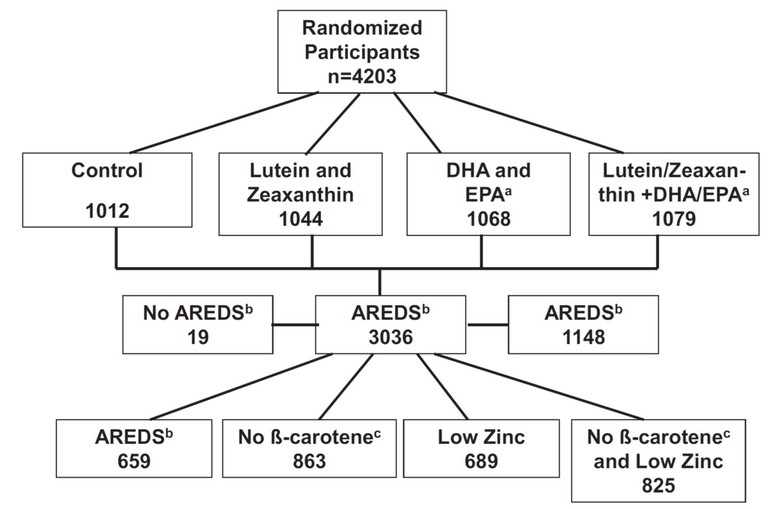
Figure 1. The AREDS2 Study Design. DHA = docosahexaenoic acid; EPA = eicosapentaenoic acid; AREDS = 500 mg vitamin C, 400 IU vitamin E, 15 mg beta-carotene, 80 mg zinc oxide and 2 mg cupric oxide. Note in the secondary randomisation, patients may have received variations of the AREDS formula (i.e., no beta-carotene, low-dose zinc, or no beta-carotene and low-dose zinc). Patients who were current smokers or had ceased smoking <1 year ago were randomised to treatments with no beta-carotene.33
1. Control (original AREDS formula),
2. Lutein/zeaxanthin only (L+Z) plus original AREDS formula,
3. DHA + EPA plus original AREDS formula, or
4. A combination of L+Z plus DHA+EPA plus original AREDS formula.
A secondary goal of the study was to see if modification to the original AREDS formulation (specifically, eliminating beta-carotene and lowering the dose of zinc) affected efficacy. The distribution of patients in the AREDS2 study is shown in Figure 1. Note there was no true placebo in AREDS2 as all participants in the study (including the ‘control’ group) received the original AREDS formulation or a modified AREDS formula based on the secondary randomisation.
The results of the AREDS2 study were somewhat surprising. It was found in primary analyses that neither DHA/EPA supplementation nor lutein/zeaxanthin supplementation added any additional protective benefit above and beyond the original AREDS formulation.24 In further sub-analyses, the use of omega 3 supplements still did not demonstrate any significant impact on AMD progression or visual acuity. This was contradictory to the previously presented data that high-dose DHA and EPA may lower the risk of AMD progression. However, the results from the AREDS2 study may have been impacted by the lack of a true ‘placebo’ group; that is, concurrent use of the AREDS supplements may have masked any further protective effect from omega-3.4,24,44 Additionally, incorrect dosage of DHA and EPA and/or insufficient duration of use may have influenced results.24 The AREDS2 study did, however, reveal several important findings, as summarised below.
LUTEIN AND ZEAXANTHIN VS BETA-CAROTENE
AREDS2 showed that the elimination of beta-carotene had no statistically significant impact on progression to late AMD.24 The AREDS2 study precluded anyone who was a current smoker or had stopped smoking less than a year before the start of the study from receiving beta-carotene. Despite this, there was still a notably higher incidence of lung cancer in former smokers who received beta-carotene.24 On the other hand, there appeared to be no increased risk of lung cancer in former smokers who received lutein and zeaxanthin.24
Additionally, the use of lutein/zeaxanthin supplements in patients with the lowest dietary intake of lutein and zeaxanthin showed a greater protective effect against late-stage AMD.24,45 There was also some thought that patients who received multiple carotenoids (that is, beta-carotene together with lutein/ zeaxanthin) as part of the AREDS2 study could have been subject to competitive binding and absorption between lutein/zeaxanthin versus beta-carotene – specifically, serum levels of lutein and zeaxanthin were lower in subjects who also received beta-carotene as part of their randomisation.32

Table 2. Australian Therapeutic Goods Administration (TGA) restrictions on zinc when used internally.28,29
To further investigate the potential benefit of lutein/zeaxanthin, a prespecified secondary analysis of data was performed in which the AREDS2 study cohort was simply split into two sub-groups – those who received lutein and zeaxanthin in conjunction with the AREDS formulation minus beta-carotene, versus those who received the original AREDS formula (including beta-carotene but no lutein/zeaxanthin). The results of this head-to-head comparison were published in 2013 in the AREDS2 Report No 3.44 They showed a favourable effect for the use of lutein/zeaxanthin instead of beta-carotene (reduction of hazard ratio by 18%). This 18% reduction was incremental to the 25% reduction seen in the original AREDS study. Given these positive findings, and the improved safety profile of lutein/zeaxanthin, the AREDS2 Research Group concluded that lutein/zeaxanthin were suitable alternate carotenoid agents that could safely replace beta-carotene in the AREDS2 formulation.45
LOW-DOSE ZINC VS HIGH-DOSE ZINC
As previously mentioned, the use of high-dose zinc (80 mg daily) in the original AREDS formula raised health concerns. AREDS2 found that reducing zinc from 80 mg to 25 mg had no statistically significant impact on progression to late AMD.24 As seen in Table 2, TGA specifically prohibits formulations with daily doses of zinc exceeding 50 mg, and those >25 mg but <50 mg must include a mandatory warning statement on the medicine label. Therefore, Table 3 summarises the AREDS2 low zinc formula that would be acceptable and suitable for use within Australia.
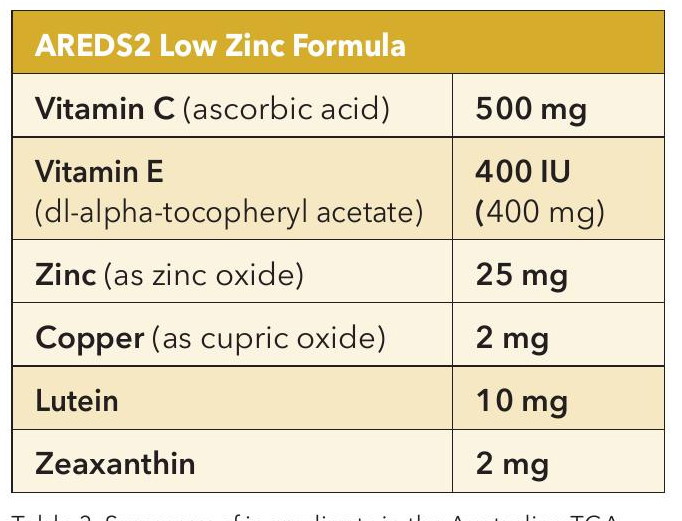
Table 3. Summary of ingredients in the Australian TGAcompliant AREDS2 low-dose zinc antioxidant formulation.
RECENT UPDATES IN AREDS2 DATASETS
Following the release of the AREDS2 data in 2013, several notable follow-up and post-hoc studies have been conducted. A long-term epidemiologic follow-up study conducted over 10 years by the AREDS2 Research Group was published in 2022 (AREDS Report 28).25 At the conclusion of the five years of the AREDS2 study, 2,923 surviving and willing participants were contacted by telephone every six months for a further five years and asked about any new diagnosis of lung cancer or progression to neovascular AMD. Additionally, 709 participants were asked to return for a clinical examination at the end of 10 years. Lung cancer incidence in the lutein/ zeaxanthin cohort remained almost half that of the beta-carotene cohort.25 Moreover, based on clinical examination and number of new diagnoses of neovascular AMD, there was also a tendency for lutein/zeaxanthin to provide an incremental increase in prevention of progression to late AMD by a further 10– 20%.25 Thus, the AREDS2 formula appeared to remain “safe and effective, even for long-term use” in patients with intermediate AMD (or late AMD in one eye only).25
More recently, a significant post-hoc analysis investigated the effect of antioxidant supplementation specifically on GA progression, and the results were published in 2025 by National Eye Institute researchers in the United States. This was a growing area of interest among clinicians given the large number of patients impacted by GA (estimated to be around five million worldwide) and the lack of a definitive treatment for GA.17 Historically, GA progression rates range from 0.53 to 2.6 mm2 /yr (median 1.78 mm2 /yr)46 and can vary between people for many reasons, including but not limited to genetics (presence of high-risk genetic variants),47 presence of reticular pseudodrusen (RPD),47,48 and other high-risk structural biomarkers.49 Additionally, the location, size, and circularity index of GA lesions also affects progression – multifocal, irregular, large, and non-central lesions tend to progress faster than their counterparts.50 Fortuitously, incident GA tends to be more commonly located non-centrally than centrally, and it also generally progresses into the central macula slower than into the peripheral macula, giving rise to a phenomenon known as ‘foveal sparing’.17
The post-hoc analysis pooled data from both the AREDS and AREDS2 datasets – 1,602 eyes were identified as having GA, of which 1,001 (62.5%) were non-centre involving.17 Progression of GA was studied using colour fundus photography, and researchers examined both total growth of GA area and “proximity-based progression… toward the central macula”.17 From a functional point of view, progression of GA towards the central macula was arguably more clinically important than the change in total GA area – small areas of non-central GA often do not impact significantly on a patient’s visual acuity, however large areas of centre-involving GA can have a markedly deleterious effect on functional vision.
Post-hoc analysis of the combined AREDS and AREDS2 data showed that high-dose antioxidant vitamins and macular carotenoids significantly slowed the progression of GA towards the fovea.17 In eyes with non-central GA, the original AREDS supplement formula (vitamins C, E, beta-carotene, plus zinc and copper) reduced the rate of GA extension into the central macula by roughly 36% compared to no supplement. In AREDS2 (recalling that all participants received the base AREDS formula and then a secondary randomisation, as per Figure 1), those further randomised to lutein and zeaxanthin experienced a further 35% slowing of GA progression and a slower rate of visual acuity decline compared to those who did not receive lutein/zeaxanthin. The researchers concluded that the effects of the AREDS antioxidants and added lutein/ zeaxanthin were complementary and additive, together slowing down the progression of non-central GA towards the fovea by approximately 55% over three years.51
Importantly, this protective effect was evident only when GA had not yet encroached on the fovea – once GA had extended to involve the foveal centre, the use of supplements conferred little-to-no benefit.17 Similarly, the use of AREDS2 antioxidant supplements had no statistically significant impact on total growth of GA area; rather, it reduced proximity-based progression towards the central macula. This led Keenan and colleagues to conclude that AREDS2 antioxidant supplements selectively slowed atrophy towards the central macula by augmenting the underlying concept of ‘fovealsparing’.17 Biologically, this makes sense: lutein and zeaxanthin are highly concentrated in the macula and supplementation with these carotenoids likely confers a protective effect.
As previously mentioned, there are now also TGA-approved intravitreal complement inhibitors available within Australia and patients with GA therefore have the benefit of multiple treatment options that may help to slow down progression. Interestingly, post-hoc analyses of two Phase 3 clinical trials of pegcetacoplan (OAKS and DERBY) failed to replicate the potential benefit of AREDS2 antioxidant supplementation in slowing proximity-based GA progression.51 This may have been due to differences in population characteristics (there was no pre-specified randomisation of antioxidant use in the OAKS and DERBY patient cohorts), imaging modality (use of colour fundus photography versus fundus autofluorescence), analytic methodology, and trial design. Recalling that AREDS2 antioxidant supplementation has been well-established to slow progression to choroidal neovascularisation, there is still benefit in recommending AREDS2 supplements to patients with intermediate AMD. However, GA remains a complex area of interest with respect to both progression and treatment options, and our understanding will undoubtedly expand as further research emerges.
AREDS2 SUPPLEMENTS IN EARLY AMD
As previously mentioned, the original AREDS study had found that the risk of progression to late AMD was fairly low in AREDS category 1 (no AMD) and AREDS category 2 (early) AMD, although the follow-up period of five years was too short to elicit a statistically significant reduction.18 Subsequently, the cohort in AREDS2 consisted of patients who were at high risk of progression to advanced AMD – that is, those with bilateral large drusen, bilateral non-central GA, or large drusen/non-central GA in one eye and late-stage AMD in the fellow eye.32 Essentially, this included patients with category 3 (intermediate) AMD in both eyes, or category 4 (late/advanced) AMD in one eye only. Thus, the benefit of AREDS2 antioxidant supplements in earlier stages of AMD remains uncertain, although use of these is unlikely to confer any harm.12,20
Interestingly, in its post-hoc analysis, the AREDS2 Report No 3 found that the use of AREDS2 antioxidant supplements (with lutein/zeaxanthin) in patients with category 3 (intermediate) AMD in both eyes slowed down disease progression by an additional 24%, versus 9% in those who already had category 4 (late/advanced) AMD in one eye.45 This indicated that the benefit from AREDS2 antioxidant supplements is significantly greater for patients who start taking these supplements earlier, and this benefit is skewed towards reduction in the risk of choroidal neovascularisation.45 It is, therefore, important for Australian optometrists to accurately grade AMD patients using a suitable classification system, and to identify, as early as possible, the correct time to commence ARED2 antioxidant supplements.
As with all AMD patients, emphasis should be placed on the importance of lifestyle modifications, such as cessation of smoking and performing light physical activity.11 Additionally, post-hoc analysis of AREDS and AREDS2 food frequency questionnaires identified that increased dietary intake of omega 3 LCPUFA (DHA/EPA), carotenoids (beta-carotene, lutein, and zeaxanthin), vitamins A, B6, B9 (folate) and C, and minerals like magnesium, may be associated with reduced risk of progression to late AMD, particularly large drusen development and GA (as opposed to neovascular AMD).14 Conversely, high dietary intake of saturated fatty acid, mono-saturated fatty acid, and oleic acid, was associated with increased risk of late-stage AMD.14
ADHERENCE TO AREDS2 ANTIOXIDANT SUPPLEMENTS
Adherence to AREDS2 antioxidant supplement use remains a critical factor, both with respect to marketing and patient use. The TGA specifically prohibits Australian brands from referencing ‘AREDS’ or ‘AREDS2’ on product packaging; rather, a practitioner is responsible for identifying patients who would benefit from antioxidant supplementation and recommending a specific product and appropriate dosing. A study conducted by Ng and Goggin52 in 2006 explored compliance with the use of an antioxidant supplement requiring an off-label practitioner recommendation of twice-daily dosing to deliver adequate supplementation (whereas on-label indicated once daily). They found that only 1% of patients were compliant with this; 95% of patients continued to follow the recommended dosing of one tablet daily. Of concern, this meant a significant portion of patients were not receiving the full benefit of AREDS supplementation in slowing down progression of AMD.
Thus, Australian optometrists play a pivotal role in educating patients, even those with long-term disease, on the availability of AREDS2 antioxidant supplements (some studies have identified that only half of suitable AMD patients are even aware of antioxidant supplements52 ), their benefits, and appropriately identifying the correct time to commence these. Optometrists should be aware of the ingredients and dosages within various AREDS2 antioxidant supplements, and be comfortable in recommending a specific brand or product that is consistent with the TGA-compliant AREDS2 low-dose zinc formulation as previously detailed in Table 3.
CONCLUSION
The incidence and prevalence of AMD is known to be increasing in line with the ageing population in Australia and the Western world. Research has shown that a combination of lifestyle changes (such as cessation of smoking and light physical activity), dietary modifications (following a Mediterranean-style diet and increasing consumption of fish, nuts, vegetables, and fruits), and appropriate recommendation of AREDS2 antioxidant supplements can play an important role in AMD patients. More specifically, both original and post-hoc results from the large-scale population-based AREDS2 study strongly suggest that antioxidant and carotenoid supplements are indicated in patients with intermediate AMD, and that these help to slow progression to late AMD (choroidal neovascularisation and centre-involving geographic atrophy).
Optometrists are in a unique position to educate patients on AMD and, as part of a holistic approach, recommend specific use of AREDS2 antioxidant supplements appropriately. Within the Australian context, patients with intermediate AMD or non-central GA should be recommended the TGA-compliant AREDS2 low zinc formula (vitamin C 500 mg, vitamin E 400 IU, copper 2 mg, zinc 25 mg, lutein 10 mg, zeaxanthin 2 mg) to slow progression to late AMD. As summarised by Tiarnan Keenan, a researcher at the National Eye Institute: “We’ve known for a long time that AREDS2 supplements help slow the progression from intermediate to late AMD… antioxidant supplementation is a simple step that may slow central vision loss, even for those with late disease.”53
To earn your CPD hours from this activity, scan the code or visit mieducation.com/areds2-nutritional-supplements-foramd-where-are-we-in-australiain-2026.

References available at mieducation.com.

Inez Hsing BAppSc (Optom) (Hons) GradCertOcTher OACAP-G is a clinical optometrist practising in Brisbane, with special interests in ocular disease and provision of collaborative care in the areas of retinal/ macular disease and glaucoma. She is a long-serving sessional academic in the Queensland University of Technology School of Clinical Sciences (Optometry and Vision Science) and currently serves on the Glaucoma Australia Board of Directors.