mitechnology
Imaging in Geographic Atrophy
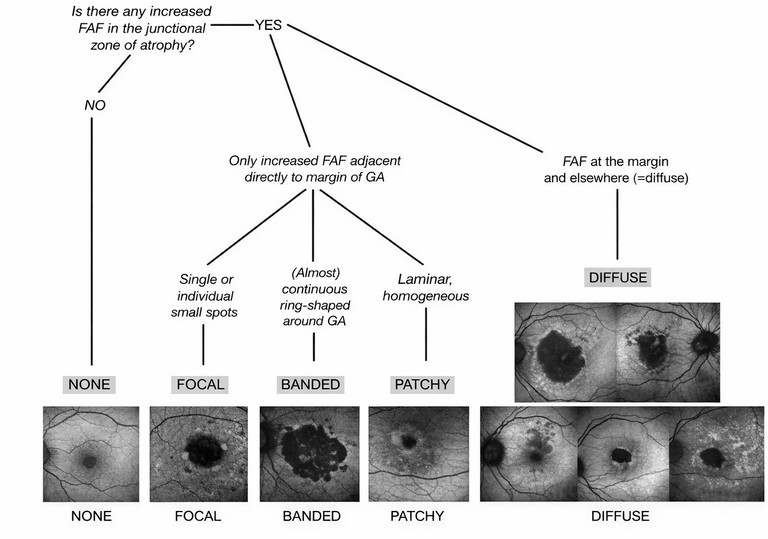
Figure 1. Perilesional FAF patterns indicative of GA lesion growth.12 Image courtesy of Creative Commons.
Geographic atrophy (GA) is a late stage of age-related macular degeneration (AMD) defined by progressive loss of retinal pigment epithelium (RPE), photoreceptors, and the underlying choriocapillaris. Because central visual acuity can remain relatively preserved until the fovea is involved, longitudinal imaging – especially lesion area and growth – has become the core objective measure for prognosis and therapeutic monitoring.1
In this article, Adam Hamilton summarises how modern imaging links metabolic stress signals, most notably fundus autofluorescence (FAF) as a surrogate of bisretinoid/lipofuscin-related RPE metabolism,2 with structural change on optical coherence tomography (OCT), including photoreceptor band disruption, RPE attenuation, and hypertransmission into the choroid.3 Additionally, it explores key modalities (FAF and OCT), practical quantification workflows including Heidelberg Engineering’s RegionFinder,4,5 and emerging artificial intelligence-based tools for segmentation, tracking, and growth prediction from OCT and FAF.6-8
WRITER Adam Hamilton
GA is a leading cause of irreversible central vision loss in older adults. Lesions usually begin parafoveally and expand over years, often coalescing and eventually involving the fovea.1 Progression rates vary widely between individuals; therefore, imaging is essential for defining phenotype, measuring growth, and providing patient-specific counselling.
Multimodal imaging is not redundant – it is complementary. FAF excels at outlining atrophic borders and perilesional ‘risk’ signatures by giving part of the ‘metabolic’ picture, while OCT provides cross-sectional structural biomarkers that confirm true RPE/photoreceptor loss and reveal early outer retinal change. OCT angiography (OCT-A) can add vascular context by quantifying choriocapillaris flow impairment around lesions.9,10
METABOLIC IMAGING SIGNATURES
Direct metabolic measurements in vivo are limited in routine patient review, but several imaging signals act as clinically useful surrogates. FAF is the most established. In AMD, the dominant FAF signal reflects bisretinoid fluorophores that accumulate as lipofuscin within RPE lysosomes. These compounds arise from photoreceptor retinoid processing and can become photoreactive, linking FAF patterns to oxidative stress and RPE dysfunction.2 Accordingly, FAF is commonly interpreted as a map of RPE metabolic burden and susceptibility to atrophy expansion.
Longitudinal FAF studies support this interpretation. Distinct perilesional FAF patterns carry prognostic information: eyes with certain abnormal autofluorescence phenotypes around GA show faster enlargement than those without such patterns.11 These patterns (Figure 1) were found to be accurate predictors of lesion growth, with the banded type and diffuse type patterns having a faster growth rate (1.81 mm2 /year and 1.77 mm2 /year respectively), than those with focal patterns (0.81 mm2 /year) for example.
FAF abnormalities can also precede the appearance and enlargement of atrophy, seen as hyper-autofluorescence areas around the lesion, consistent with a sequence of metabolic stress followed by structural loss.13 Clinically, this makes FAF valuable, not only for measuring lesion size, but also for stratifying risk of progression and targeting follow-up intensity.
Near-infrared reflectance (NIR) and related near-infrared autofluorescence signals can complement blue-light FAF, particularly in patients with media opacity or when foveal-sparing disease makes certain borders difficult to appreciate. However, changes in intensity in any fundus image modality can be confounded by signal blocking or acquisition artifacts, which is why OCT correlation is important.
STRUCTURAL OCT BIOMARKERS
OCT is indispensable for GA because it directly visualises outer retinal architecture. Hallmark structural signs include thinning/ loss of the outer nuclear layer, disruption of the ellipsoid zone and other photoreceptor bands, attenuation or discontinuity of the RPE complex, and increased choroidal signal beneath areas of RPE loss (hypertransmission, Figure 2). These findings provide a structural confirmation that reduced FAF truly represents atrophy rather than masking.
OCT also supports quantitative monitoring. Spectral-domain OCT images can visualise and quantify GA area over time while simultaneously linking en face change to B-scan evidence of photoreceptor and RPE loss.3 Pragmatically, OCT helps avoid common confounding FAF factors, with outer retinal tubulation, subretinal hyperreflective material, pigment clumping, and drusenrelated shadowing all potentially altering FAF appearance without representing true atrophic expansion. By reviewing the registered B-scans, clinicians can classify ambiguous regions and maintain consistent segmentation rules across visits.
“FAF excels at outlining atrophic borders and perilesional ‘risk’ signatures by giving part of the ‘metabolic’ picture”
Finally, OCT enables structure–function conversations. Because visual acuity often underestimates functional impact until the fovea is affected, combining OCT with functional testing (e.g., microperimetry, low-luminance visual acuity) can better reflect patient experience.1
OTHER IMAGING MODALITIES
While this article focuses on OCT and FAF, as the most common imaging used in GA diagnosis and monitoring, it’s important to remember that other images can also be useful at times. These include:
OCT-A. Noninvasive choriocapillaris assessment; flow deficits around GA have been associated with progression and may add prognostic value.9,10
Colour fundus photo. Baseline documentation of overall AMD phenotype; limited sensitivity for subtle atrophic borders compared with FAF/OCT.
In research settings, adaptive optics and fluorescence lifetime imaging add mechanistic insight, but these are not available to most clinicians.
REGIONFINDER AND MEASUREMENT WORKFLOWS
Reliable lesion quantification requires more than a single good image: it depends on standardised acquisition, registration, and consistent editing rules. On the Heidelberg Spectralis platform, RegionFinder (Figure 3) provides semiautomated GA delineation on FAF by detecting areas of reduced autofluorescence and allowing clinician refinement. The RegionFinder guidance emphasises consistent acquisition and annotation practices to improve repeatability.4
Evidence comparing manual and semiautomated approaches supports RegionFinder’s utility when quality control is applied. Mahmoudzadeh and colleagues found that semiautomated measurements can perform comparably to manual approaches, while reducing workload, provided that graders use consistent rules and review images to confirm that segmentation reflects true atrophy.5 For longitudinal reporting, exporting lesion area and square-root– transformed growth metrics can reduce baseline-size dependence and align with common trial methodology.1
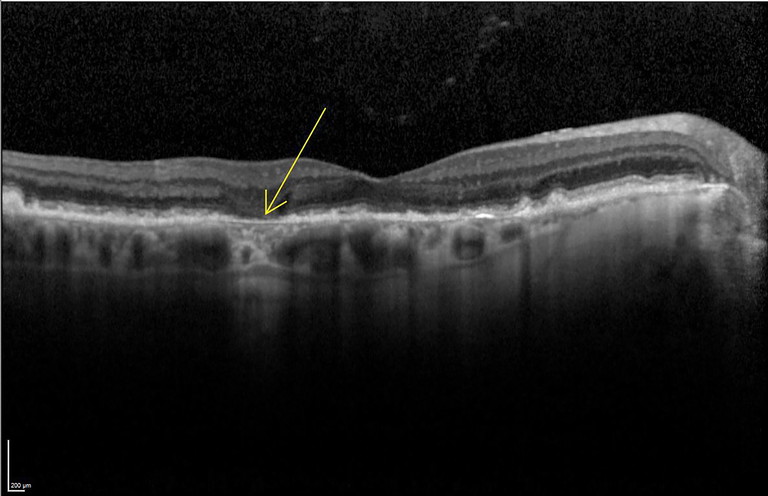
Figure 2. Example of hypertransmission. Image courtesy of Heidelberg Engineering.
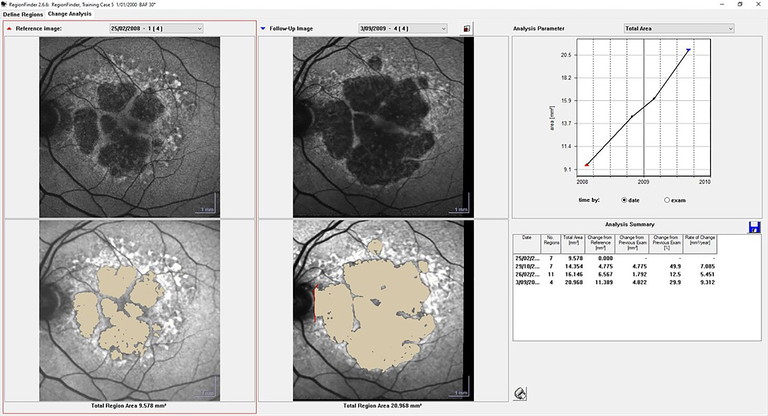
Figure 3. Example report from RegionFinder on the Spectralis platform. Image courtesy of Heidelberg Engineering.
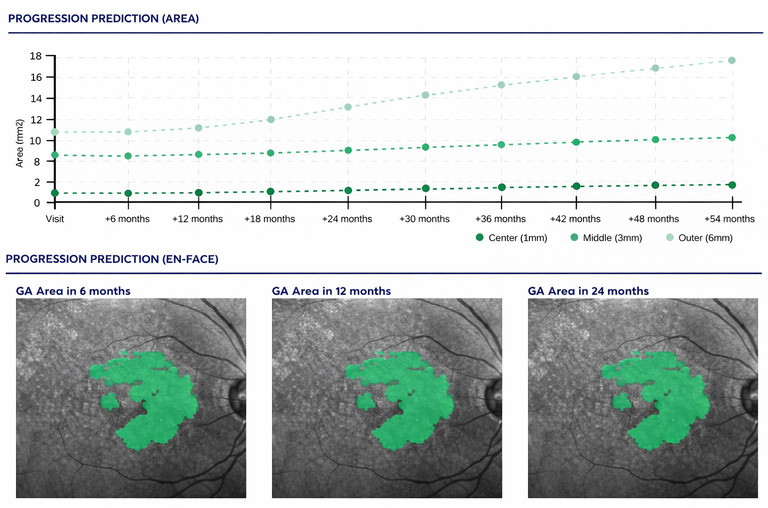
Figure 4. Example RetinAI report showing growth predictions.
“changes in intensity in any fundus image modality can be confounded by signal blocking or acquisition artifacts, which is why OCT correlation is important”
AI-ENABLED MONITORING: FROM SEGMENTATION TO PREDICTION
AI methods are increasingly positioned to standardise GA endpoints by automating segmentation, longitudinal tracking, and growth prediction. A key trend is to exploit OCT-derived representations that enhance atrophy contrast. Chu et al. demonstrated automated GA segmentation using optical attenuation coefficient (OAC) maps, suggesting that feature-engineered OCT biomarkers can improve delineation robustness.6 Complementary approaches learn directly from OCT volumes while respecting their layered structure, such as column-based deep learning methods for GA segmentation.8
Prediction is the next frontier: models aim to forecast where GA will expand, potentially generating individualised risk maps. Salvi et al. reported deep learning predictions of future GA growth from baseline FAF, illustrating that spatial growth patterns can be learned from routinely acquired imaging.7 Clinically, these systems are most useful when they integrate into review workflows (e.g., an auto-generated mask with editable boundaries), provide quality checks and uncertainty, and support device-agnostic longitudinal registration.
With companies such as RetinSight, RetinAI (Figure 4), AltrisAI, and others all available for use in research, and RetinSight registered with the Therapeutic Goods Administration and already being made use of in some clinics, the future is almost here. But as previously noted, OCT provides structural information only, not the potential for metabolic-related information from other imaging modalities.
CONCLUSION
GA imaging has become a quantitative, multimodal discipline. FAF remains central for lesion measurement and metabolic-risk phenotyping.2,11 OCT provides structural confirmation and sensitive biomarkers of outer retinal degeneration,3 and OCT-A may add prognostic vascular context through choriocapillaris flow metrics.9,10 Standardised workflows – including RegionFinder on Heidelberg platforms – translate these data into consistent longitudinal endpoints.4,5 AI tools are now poised to scale this process for structural change analysis by enabling predictive monitoring, supporting both routine care and clinical trials.6,7,8
Adam Hamilton BSc MHRM has 20 years’ experience in the ophthalmic community. Commencing his profession as a new graduate working in an ophthalmic clinic, his passion for imaging was sparked by an old Canon film camera – he would spend days in the dark doing angiograms. Ten years ago, he joined Heidelberg Engineering and currently works as Manager of Professional Education (Asia Pacific), raising awareness about the benefits and potential pitfalls of imaging technology in eye health in clinics around Asia.
References available at mivision.com.au.