mieducation

The Challenge of Neuroadaptation for the Cataract SurgeonThe Challenge of Neuroadaptation for the Cataract Surgeon
Neuroadaptation has emerged as a hot topic for cataract and refractive surgeons, as some patients are challenged by presbyopia-correcting intraocular lenses (PC IOLs) and extended depth of focus (EDOF) lenses. While most patients adapt quickly to these implants, some adapt more slowly, and a small minority not at all. This article explores this specific challenge that we face in modern cataract surgery, and how to best manage patient expectations. It also looks at ways to improve outcomes.
WRITER Dr Alex Ioannidis
LEARNING OBJECTIVES
On completion of this CPD activity, participants should be able to:
1. Understand the process of neuroadaptation,
2. Be aware that neuroadaptation post intraocular lens implant can take time,
3. Be able to explain neuroadaptation and set realistic expectations, and
4. Be aware of options should a patient fail to neuroadapt post-IOL surgery.
In a world that is ever changing, neuroadaptation is an important survival aid. The brain constantly receives thousands of stimuli from peripheral receptors – including sight, smell, sound, plus a myriad of other stimuli from internal sensors in the viscera and pressure sensors in our hands and feet.
Neuroadaptation is the ability to suppress stimuli that are non-essential at a given moment, while at the same time augmenting others that are important for survival. Additionally, it is the ability to adapt to new situations entering our consciousness – namely new stimuli that aid our existence. This requires a level of plasticity in our nervous system.
Neuroadaptation occurs at a subconscious level involving a number of brain centres. It is an active process requiring the creation of new neural pathways.
PHYSIOLOGY OF VISUAL NEUROADAPTATION
The process of visual neuroadaptation starts soon after birth and is a component of normal development. When light first hits a baby’s retina at birth, a dramatic flood of information travels to the occipital cortex. And when a baby first registers an image, the physics of optics predict that the image is inverted. Neuroadaptation flips it cortically so that up is down and down is up – this is a primordial step in the development of normal vision. At the same time, other visual functions start to develop, such as stereopsis and the ability to focus. Later in life, neuroadaptation allows for coping with changes in light and dark, and suppresses unimportant stimuli that enter our awareness.
The process of neuroadaptation is thus a process that requires active learning throughout life.
In relation to IOL surgery, functional magnetic resonance imaging (MRI) studies have shown that several brain centres are involved, including the frontal, middle frontal, parietal frontal, and postcentral gyrus. Furthermore, areas in the brain associated with learning, task planning, and problem solving become active in subjects with dysphotopic symptoms after IOL surgery.1 This indicates the complex nature of neuroadaptation and the plasticity required to adapt to a new IOL after surgery.
Patients often ask how long it takes to get used to a new IOL. The short answer is that in most cases the process can take several days to weeks. But in some rare cases it can take several months, depending on the individual patient’s brain plasticity.
One of the problems we have in modern cataract surgery is that we have no tools to measure this ability in a given patient and thus pre-empt any potential problems with a specific IOL design.
Examples of Neuroadaptation in Ophthalmology
We all employ visual neuroadaptation on a day-to-day basis in response to various visual phenomena. One example is how we perceive and adapt to entoptic phenomena in the visual system.
By definition, entoptic phenomena arise from structures within the eye. Floaters, which most of us see intermittently when looking at a white wall or the sky, are a common example. It is through the process of neuroadaptation that we can suppress the stimulus that they exert on the retina and thus, are able to go about our business, not paying much attention to them. We do, however, pay attention to a new floater and this can prompt a visit to the optometrist or ophthalmologist to exclude a retinal tear or detachment. Thus, an important feature of neuroadaptation is the response to change, i.e., to a new stimulus or change in status.
Similarly, the sudden and dramatic loss of field caused by a vascular event, such as after a branch vein occlusion or an anterior ischemic optic neuropathy (AION), will often prompt a patient to visit a health care practitioner, be it their general practitioner, optometrist, or ophthalmologist. In this example, we see how a sudden change in what we perceive through the visual system is more likely to be registered; in other words the individual becomes ‘aware’ of the change in their vision.
Patients with glaucoma often don’t realise their loss of peripheral visual field because the process is slow and insidious. The patient, in a sense, ‘neuroadapts’ to the gradual change – with the unfortunate consequence of gradual and permanent loss in visual function. In this case, neuroadaptation works against the patient due to a maladaptive response.2
Another example of neuroadaptation involving the visual system working against the individual is the gradual loss of vision that keratoconus causes. Despite having significant visual blur through irregular astigmatism and an uncorrected refractive error, many keratoconics do not present to an optometrist with a visual complaint until their condition has progressed significantly – in a sense the visual system neuroadapts to the poor image formed in the retina as the process is gradual and insidious.
MULTIFOCALITY IN MODERN CATARACT SURGERY
Currently, more cataract surgeons are turning to EDOF IOLs and PC IOLs to correct presbyopia in their patients or enhance intermediate vision. By their very design, modern EDOF/PC IOLs create a construct of physiologic binocular vision by allowing for multiple foci, that are variously focused and defocused, within a single eye. This is a novel experience for the visual system as this type of multifocality does not exist in the real world. A consequence of this is that the visual system requires a period of adjustment as there is no pre-wired circuitry with which to digest the information from this new multifocality.
According to the American Society of Cataract and Refractive Surgery (ASCRS)/ European Society of Cataract and Refractive Surgery (ESCRS) survey update of complications of foldable IOLs, the most common cause for explantation or secondary intervention after multifocal IOL surgery is the presence of disturbing glare/haloes and other optical aberrations.3
In a study of 43 eyes undergoing PC IOL implantation, 35 patients (81%) had improvement with conservative treatment or watchful waiting. Five patients (12%) did not improve despite treatment combinations, and three patients (7%) required IOL exchange due to ongoing symptoms.4 This data suggest that most patients can be treated conservatively and only a very small number may require further surgery to resolve the issue.4
Hence the period of neuroadaptation is variable and cannot be predicted, however we do know, from experience, that most patients are able to neuroadapt to a new IOL within a few weeks. This is evident from the fact that most patients report some dysphotopic symptoms after surgery for a short period, and then on subsequent visits fail to mention the glare, shadow, or flicker that was initially apparent.
In a study of 30 patients undergoing PC IOL surgery, initial areas of brain activation gradually became quiescent as the process of neuroadaptation was completed. Patients showed increased activity of cortical areas involved in visual attention, procedural learning, effortful cognitive control, and goal-oriented behaviour in the early postoperative period. This normalised by the sixth postoperative month.5
POTENTIAL BARRIERS TO NEUROADAPTATION
Probably the biggest barrier to neuroadaptation after IOL surgery is the patient’s stress response to the new visual stimulus. In other words, how the patient interprets the new stimulus as a positive or negative experience.
Patients do well when they understand that the occasional flicker or shadow after surgery is to be expected and is part of the healing or learning that is required after surgery.
Patients who fail to neuroadapt are those who become fixated with the symptom and cannot accept the stimulus as part of the process. This issue can be compounded if friends or relatives state that when they had surgery, they didn’t have the same experience (and everything was “absolutely perfect”). If this occurs, the patient is likely to assume that something has gone wrong with their operation.
Some patients will enter into a cycle of self-rumination around the issue. This is a situation that most cataract surgeons have faced, at least once in their careers, and it can be quite challenging to manage.
“Patients who fail to neuroadapt are those who become fixated with the symptom and cannot accept the stimulus as part of the process”
It is imperative to first perform a full examination to ensure that the flicker or shadowing in the postoperative period is not due to an organic cause, such as retinal traction or detachment. Once this has been ruled out, a surgeon’s gentle supportive advice, assuring the patient that things will get better in time, will generally set the patient at ease. Patients generally respond well to a caring approach rather than a dismissal that all is OK; avoid disparaging comments such as “it’s all in your head” – such an approach is more likely to lead to confrontation and will exacerbate the symptoms and dissatisfaction further.
Other barriers to neuroadaptation after IOL surgery include undiagnosed neurological conditions affecting the centres involved in the process of neuroadaptation. Some patients may have silent strokes in one or more regions of the brain that may not be known prior to surgery. Patients can fail to report a history of patching as a child (to treat amblyopia), the use of a mild prism in their glasses, or ocular trauma many years ago (subtle phorias affecting ocular motility). Things that appear as minor may affect the ability to adapt to the new IOL. A thorough history is essential so as not to miss important issues.
In a recent paper, the role of the Mini-Mental State Examination (MMSE) to assess a person’s ability to neuroadapt after cataract surgery, was investigated. It found that younger patients with lower MMSE scores adapted faster to a new IOL than older patients, and this was statistically significant. Although the MMSE is used to detect early dementia, it may have a role as a surrogate for analysing processing brain power in some patients undergoing cataract surgery, and the ability to adapt to certain types of IOL.6
IMPORTANT PREOPERATIVE CONSIDERATIONS
There are several steps one can take to reduce the risk of neuroadaptation issues after cataract surgery. Probably the most important is to identify which patients will not respond well to a given IOL. For instance, patients with existing neurological conditions, ocular dysmotility, patching as a child, and phorias, must be excluded from receiving a PC IOL as they may face issues with adaptation to the new IOL due to convergence and excessive angle kappa deviation.
Patients with very exacting personality traits – perfectionists who demand specific functional outcomes from cataract surgery – may also face issues with adaptation and need to be counselled properly as to realistic refractive outcomes.
Another cohort that needs careful consideration is those with significant ocular surface disease /dry eye. In these patients, despite meticulous management of the dry eye, transient blurring of vision is common. This is due to poor surface wetting /tear film instability and can lead to significant levels of dissatisfaction after PC / EDOF IOL surgery.
In a recent paper describing levels of dissatisfaction after EDOF IOL surgery, dry eye was the second cause of poor tolerance after residual ammetropia in 26.5% of cases.7 Patients with reported entoptic phenomena, such as floaters, are also poor candidates for EDOF and PC IOLs as the media opacities can cause glare, haloes, and other visual disturbances such as waxy vision. Patients with significant corneal opacities caused by trauma, herpes simplex virus infection, and surface degenerations, should also be counselled against having an EDOF or a PC IOL. These patients may have significant irregular astigmatism that will not be corrected, despite the use of a toric IOL, and may complain of a residual refractive error and poor vision.
Other optical considerations that have been reported to increase the levels of dissatisfaction after surgery include high angle kappa, high myopia, significant astigmatism, large preoperative pupil size, and reduced central corneal thickness.8

Figure 1. The patient complained of an oblique line in her vision. This was caused by an oblique line in the capsular bag. She was unable to neuroadapt to this stimulus and was concerned about her vision.

Figure 2. Post YAG capsulotomy, the symptoms resolved and the patient was happy to undergo a second eye cataract surgery.
Case 1: “I Have a Line in My Vision”
Mary Davis,* a 65-year-old female patient, underwent routine cataract surgery. Her postoperative vision was 6/6 unaided and she was pleased with the clarity of vision. She reported, however, an oblique line in her vision that was constant. On examination she was found to have a subtle fold of the posterior capsule (Figure 1). She was quite concerned as she was due to have her second eye done two weeks later. In a case like this, it is imperative to resolve the issue in the first eye prior to operating on the second eye, as the patient is unlikely to neuroadapt. Ms Davis was therefore treated with a YAG capsulotomy and the issue was resolved (Figure 2). She was then happy to proceed with surgery to her other eye as originally planned.
Ms Mable presented with diagrams she had drawn of the glare and haloes around headlights of cars and streetlamps (Figures 3 and 4). This patient clearly had been unable to neuroadapt to the PC IOLs implanted. She was happy with her distance and reading vision, but she found it difficult to go out after dusk due to the severe dysphotopsia. She made it quite clear that she did not want any more surgery. We discussed the option of using pilocarpine 2% in the evenings to reduce pupillary size, to see if the dysphotopsia was reduced. The trial with this medication proved helpful and she was able to start going out in the evenings with intermittent use of pilocarpine 2%, one hour prior to departure (Figures 5 and 6).
The use of miotics to treat dysphotopsia has been described in the literature and is a useful modality to resolve issues with neuroadaptation in some patients. Apart from pilocarpine, brimonidine tartrate has also been used in some settings and its miotic effect is well documented.9
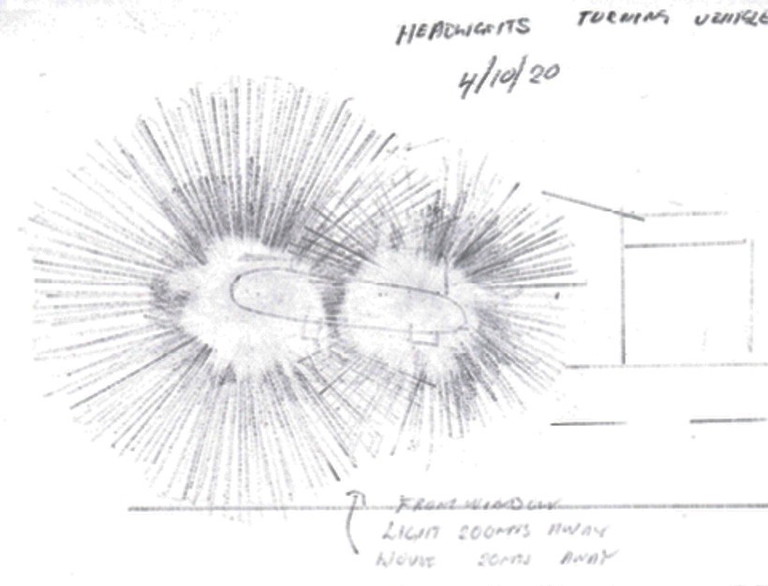
Figure 3. Drawing of car headlight and resultant glare, sketched by the patient and presented at her first consultation.
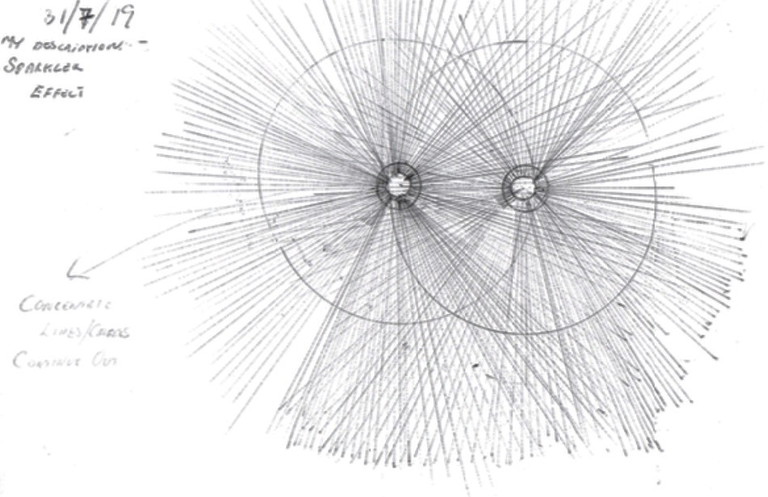
Figure 4. Car headlights head on. Severe glare and haloes are depicted in this sketch by the patient.
“… an important feature of neuroadaptation is the response to change, i.e., to a new stimulus or change in status”
Case 2: “I Cannot Go Out at Night”
Alice Mable,* a 67-year-old female patient, presented having seen a number of ophthalmologists complaining of glare and haloes after PC IOL surgery two years previously by another surgeon. She had received bilateral diffractive IOL implants. Her acuities were 6/5 unaided in both eyes and she was reading N5 unaided comfortably. Her complaint was severe dysphotopsia, and she was unable to go out at night due to significant glare and haloes with car headlights.
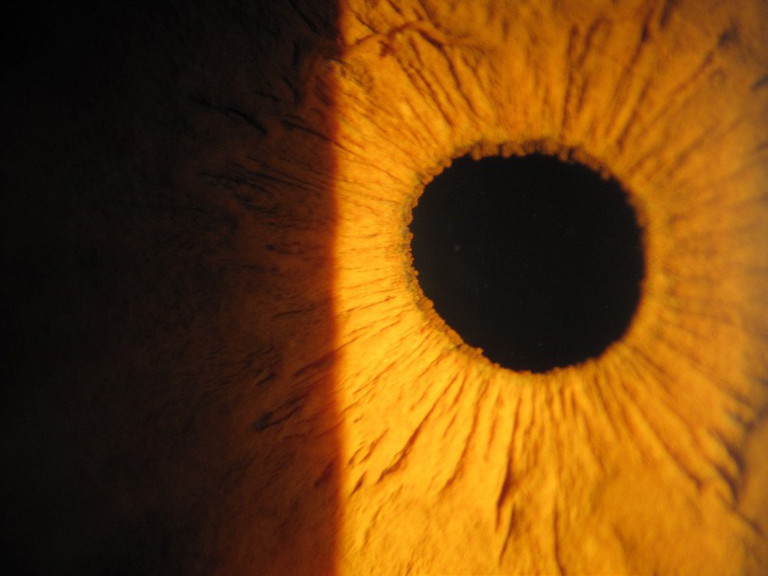
Figure 5. Miosed pupil in a patient intolerant to a diffractive PC IOL. This patient presented for a second opinion. She was very happy with the refractive outcome as her vision was 6/5 UA in both eyes and N5 for near, but was unhappy with the quality of the vision (described as hazy).

Figure 6. The effect of pupillary miosis is to reduce the optical effect of the diffractive rings (echellettes) on the IOL ( black arrow). The patient was happier with the quality of the vision and the use of pilocarpine 2% at night.
MANAGEMENT OPTIONS WHEN NEUROADAPTATION FAILS
Despite our best efforts, on occasion patients will fail to neuroadapt to their new IOL within a short time after surgery. It is very important, in this instance, to undertake a full examination to exclude issues with the IOL such as surface damage, cracks or other defects that can induce photic phenomena (Figure 7).
It is also important to examine the retina, with full dilatation of the pupil, to exclude any retinal pathology such as detachment or tears. In most cases the examination will be entirely normal, which confirms that we are dealing with a case of failure to neuroadapt to the new IOL.
Often these patients will have very good vision but complain of glare, haloes or other symptoms, such as streaks of light or shadows in the periphery (negative dysphotopsia).
Once the examination is completed, it is helpful to explain the process of neuroadaptation to the patient in simple words. It is important to emphasise that the process can, in some cases, take a bit of time as the brain learns to accept the new IOL – the key point is that the brain needs time to adjust to the new implant and that the visual symptoms will gradually fade away.
Studies have shown that the levels of stress in patients with an ophthalmic condition can be very high and can reach levels similar to having been told of a diagnosis of cancer.10 However, a careful explanation is often all that is needed to allay any fears that there has been a complication with the patient’s operation. Hence it is important not to be dismissive, and to allow patients to express their concern about what they are experiencing. Another important point is to arrange further follow up to review how things are progressing four to six weeks later. In many instances, the patients don’t mention the original complaint – hence indicating that the process of adaptation has occurred.
On the very rare occasion that a patient still complains of visual symptoms, a trial of miotics can prove helpful. If this also fails, then the final option is to remove the IOL and exchange it with a different IOL design. As this is the final option, patients in this situation must be counselled appropriately as to the final refractive outcome.
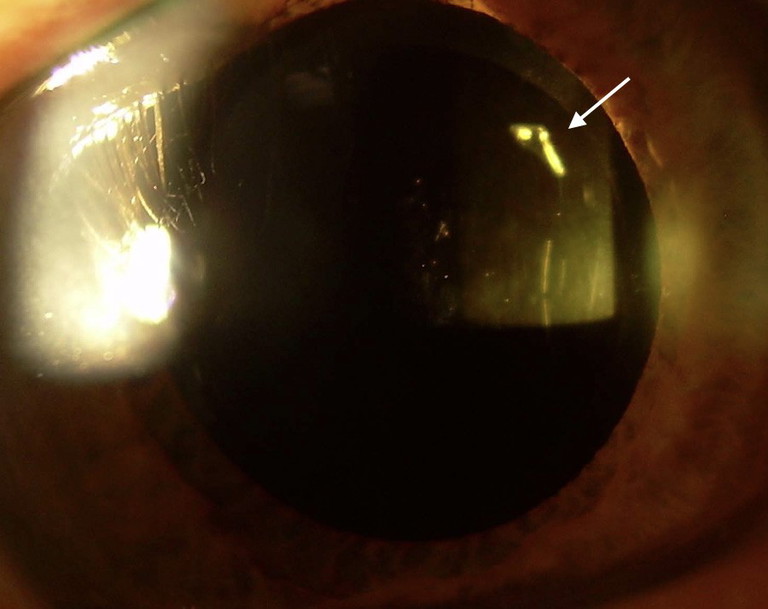
Figure 7. In this case, the IOL had a small defect (white arrow) causing dysphotopsia with poor neuroadaptation due to the symptoms of a light flicker. The IOL was explanted and the issue resolved.
OTHER CHALLENGES
There are several additional challenges that we face with emerging IOL technology. For financial and proprietary reasons, companies developing new IOLs cannot divulge how their new lens technology ‘works’ to create the presbyopia correcting effect, which makes it difficult for surgeons to understand and best convey information to their patients. Furthermore, the nomenclature and concepts in optics can be difficult to understand, particularly for elderly patients who have no education in optics and refraction. It is not, therefore, unusual for patients to default to “I will do what you recommend, Doc”, but this can lead to misunderstandings or second thoughts on outcomes later.
CONCLUSION
We live in a time of huge advances in IOL technology and ophthalmologists have a wide range of options to improve outcomes in cataract surgery, including presbyopia correction. This variety of options does bring its own sets of problems as some patients can find it difficult to decide on what they want to achieve as a functional visual outcome.
It is, however, our job to explain as best we can, the challenges of vision and perception, and how unique that is to every individual – there is no such thing as the best IOL but rather a set of options – each with its own limitations and compromises.
It is fortunate that in most cases, patients do come to understand these complex concepts and are willing to accept the options available with appropriate guidance from their practitioner.
Medicine is both an art and a science. The science has provided us with a wide range of new IOLs to help improve vision and achieve presbyopia correction – the art is in how we guide our patients to make the best possible decision about the IOL they are about to receive.
*Patient names changed for anonymity.
To earn your CPD hours, scan the code or visit mieducation. com/the-challenge-ofneuroadaptation-for-thecataract-surgeon.

“… there is no such thing as the best IOL but rather a set of options – each with its own limitations...”
References
1. Rosa AM, Miranda ÂC, Castelo-Branco M, et al. Functional magnetic resonance imaging to assess the neurobehavioral impact of dysphotopsia with multifocal intraocular lenses. Ophthalmology. 2017 Sep;124(9):1280-1289. doi: 10.1016/j. ophtha.2017.03.033.
2. Crabb DP, Smith ND, Garway-Heath DF, et al. How does glaucoma look?: Patient perception of visual field loss. Ophthalmology. 2013 Jun;120(6):1120-6. doi: 10.1016/j. ophtha.2012.11.043.
3. Mamalis N, Brubaker J, Werner L, et al. Complications of foldable intraocular lenses requiring explantation or secondary intervention – 2007 survey update. J Cataract Refract Surg. 2008 Sep;34(9):1584-91. doi: 10.1016/j. jcrs.2008.05.046.
4. Woodward MA, Randleman JB, Stulting RD. Dissatisfaction after multifocal intraocular lens implantation. J Cataract Refract Surg. 2009 Jun;35(6):992-7. doi: 10.1016/j.jcrs.2009.01.031.
5. Rosa AM, Miranda ÂC, Murta JN, et al. Functional magnetic resonance imaging to assess neuroadaptation to multifocal intraocular lenses. J Cataract Refract Surg. 2017 Oct;43(10):1287-1296. doi: 10.1016/j.jcrs.2017.07.031. 6. Nishi Y, Nishi H, Nishi O, et al. Clinical assessment of brain adaptation following multifocal intraocular lens implantation. BMC Ophthalmol. 2025 Mar 7;25(1):116. doi: 10.1186/s12886-025-03955-7.
6. Nishi Y, Nishi H, Nishi O, et al. Clinical assessment of brain adaptation following multifocal intraocular lens implantation. BMC Ophthalmol. 2025 Mar 7;25(1):116. doi: 10.1186/s12886-025-03955-7.
7. Wanten JC, Bauer NJC, Nuijts RMMA, et al. Dissatisfaction after implantation of EDOF intraocular lenses. J Cataract Refract Surg. 2025 May 1;51(5):399-405. doi: 10.1097/j. jcrs.0000000000001615.
8. Jeon W, Yoon CH, Kim MK, et al. Risk factors in self-reported dissatisfied patients implanted with various presbyopia-correcting intraocular lenses after cataract surgery. BMC Ophthalmol. 2025 Feb 19;25(1):82. doi: 10.1186/s12886-025-03912-4.
9. Marques FF, Marques DM. Unilateral dysphotopsia after bilateral intraocular lens implantation using the AR40e IOL model: Case report. Arq Bras Oftalmol. 2007 Mar- Apr;70(2):350-4. doi: 10.1590/s0004-27492007000200030.
10. Williams RA, Brody BL, Brown SI, et al. The psychosocial impact of macular degeneration. Arch Ophthalmol. 1998 Apr;116(4):514-20. doi: 10.1001/archopht.116.4.514.

Dr Alex Ioannidis MBBS FRCOphth FRANZCO is an ophthalmic surgeon with over 20 years of experience performing small incision refractive cataract surgery. He practises at the Vision Eye Institute, Retina Specialists Victoria, and Mornington Specialist Eye Clinic, where he also offers tailored surgical solutions for his patients with conditions of the anterior segment. As an expert in his field, he has published extensively and has been the recipient of several awards for his research and academic achievements.