mieducation

Managing Dry AMD In Conversation with Dr Anthony Kwan
While there is no established treatment currently available for dry age-related macular degeneration (AMD), we now have treatments in Australia for geographic atrophy (GA). Optometrists, as primary eye health providers, play a crucial role in the diagnosis of patients with these two diseases, providing them with education, support, and ongoing monitoring under shared care relationships with ophthalmology. To prepare this CPD article, optometrist Tina Huynh interviewed Associate Professor Anthony Kwan, an academic retinal specialist from the Queensland Eye Institute in Brisbane, about recognising AMD and early GA on imaging, when and how to refer for complement inhibitor treatment, and how to explain benefits, risks, cost, and long-term commitment to patients.
WRITERS Associate Professor Anthony Kwan and Tina Huynh
LEARNING OBJECTIVES
On completion of this CPD activity, participants should:
1. Be able to identify key clinical and imaging signs of dry AMD and GA and know when ophthalmology referral is indicated,
2. Understand how intravitreal complement inhibitors work, who benefits most, and what trial data show,
3. Be able to clearly set expectations, support adherence, and involve patients in shared decisions, and
4. Implement a shared care model for monitoring, co-managing risk factors, coordinating referrals, and supporting vision rehabilitation when needed.
DIAGNOSING DRY AGE-RELATED MACULAR DEGENERATION IN PRACTICE
Q. In an optometry practice with a retinal camera and optical coherence tomography (OCT), what are the key features that assist in the diagnosis of dry age-related macular degeneration (AMD)?
On colour fundus photos, dry AMD starts with drusen and subtle pigment changes at the macula – small yellow deposits and areas of mottling under the retina before any obvious scar appears.1,2 Larger, soft, confluent drusen and clusters, especially with retinal pigment epithelium (RPE) hyper- and hypopigmentation, signal intermediate disease and a higher risk of progression to late AMD, including GA.1-3
On OCT, look for elevation and thickening of the RPE from drusen, disruption of the ellipsoid zone, and any early outer retinal and RPE thinning that suggests nascent atrophy.3-5Reticular pseudodrusen (subretinal drusenoid deposits) are important risk markers, as they are associated with faster progression to advanced AMD, including GA.4,5 Fundus autofluorescence (FAF), where available, acts like a ‘heat map’ – areas of mixed increased and decreased autofluorescence around drusen or early atrophy often mark zones where GA will later expand.3-5
One way to explain this to patients: “Dry AMD is a bit like rust developing under the paint of a car bonnet – drusen are the rust bubbles, and our scans (photos, OCT, autofluorescence) show where the paint is starting to lift before a full hole (atrophy) appears (Figures 1–3).”2,6
DEFINING AND MONITORING EARLY GEOGRAPHIC ATROPHY
Q. What is considered early GA, and how should optometrists monitor these patients before referral?
GA is essentially a sharply defined ‘bald patch’ at the macula, where photoreceptors, RPE, and often choriocapillaris, have disappeared, usually evolving from areas of drusen and pigment change in non-exudative AMD.5,7 Early GA often refers to smaller lesions, usually extrafoveal, or to nascent GA (nGA) on OCT: localised RPE attenuation, hypertransmission into the choroid, and loss of outer retinal layers before the fovea is fully involved.4,5,7
In primary care, if GA is small, non-foveal, and the patient is asymptomatic or minimally symptomatic, guidelines suggest optometric review around every six to 12 months, sooner if there are high-risk features or advanced disease in the fellow eye.2,8 Monitoring includes repeat OCT and, where possible, FAF to document lesion size and growth, reinforcing home monitoring (Amsler, reading tasks), and aggressive risk-factor management: smoking cessation,
AREDS2 supplementation where indicated, cardiovascular risk control, and optimising lighting and magnification.1,2,8
A patient-friendly analogy: “Imagine a healthy lawn: early GA is like a small bare patch off to the side – the grass there has died, but you still walk fine across most of the yard; our job is to watch that patch closely and slow any spread towards the front gate you use every day (your central vision).”7
CURRENT TREATMENT OPTIONS
Q. What are the current treatment options available for dry AMD patients?
For early and intermediate dry AMD, the mainstays remain lifestyle and systemic risk-factor control, evidence-based supplementation, and visual optimisation – these interventions still make the greatest impact at the population level.1,2,8 That means smoking cessation, cardiovascular risk optimisation, a diet rich in leafy greens and fish, AREDS2 supplements for the appropriate phenotypes, and timely low-vision or rehabilitation referral when function is impaired.1,2,8
For GA, the major change is intravitreal complement inhibitor therapy – pegcetacoplan (a C3 inhibitor, Syfovre) and avacincaptad pegol (a C5 inhibitor, Izervay) – both approved in the US and more recently registered by the Therapeutic Goods
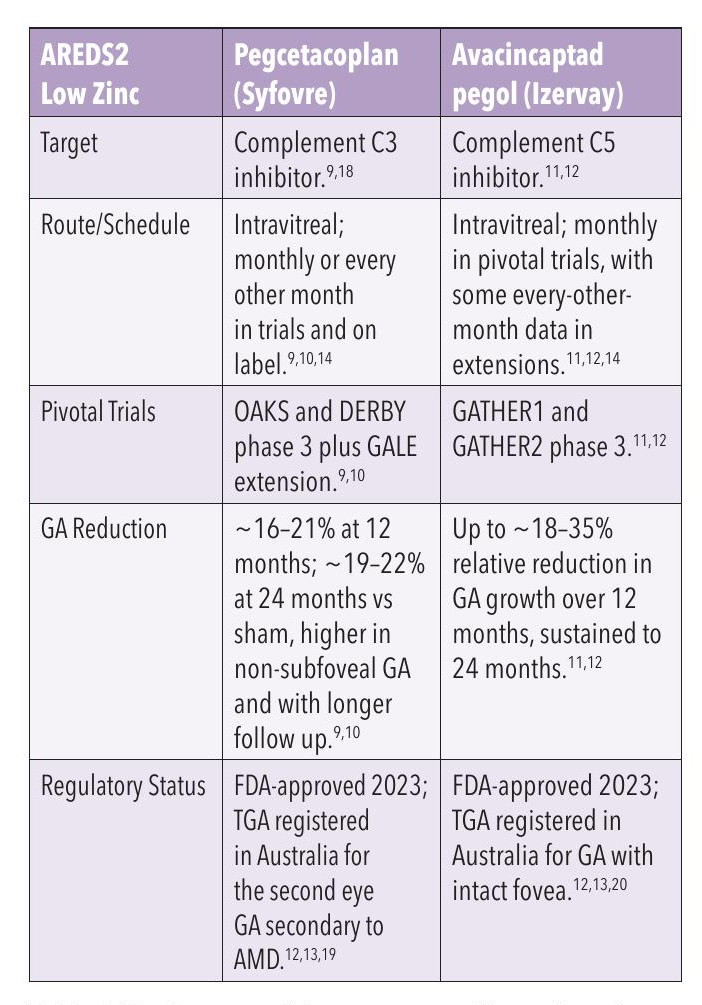
Table 1. Key features of the two agents (Note: there is no head-to-head study between the molecules, this is for illustrative purposes only).
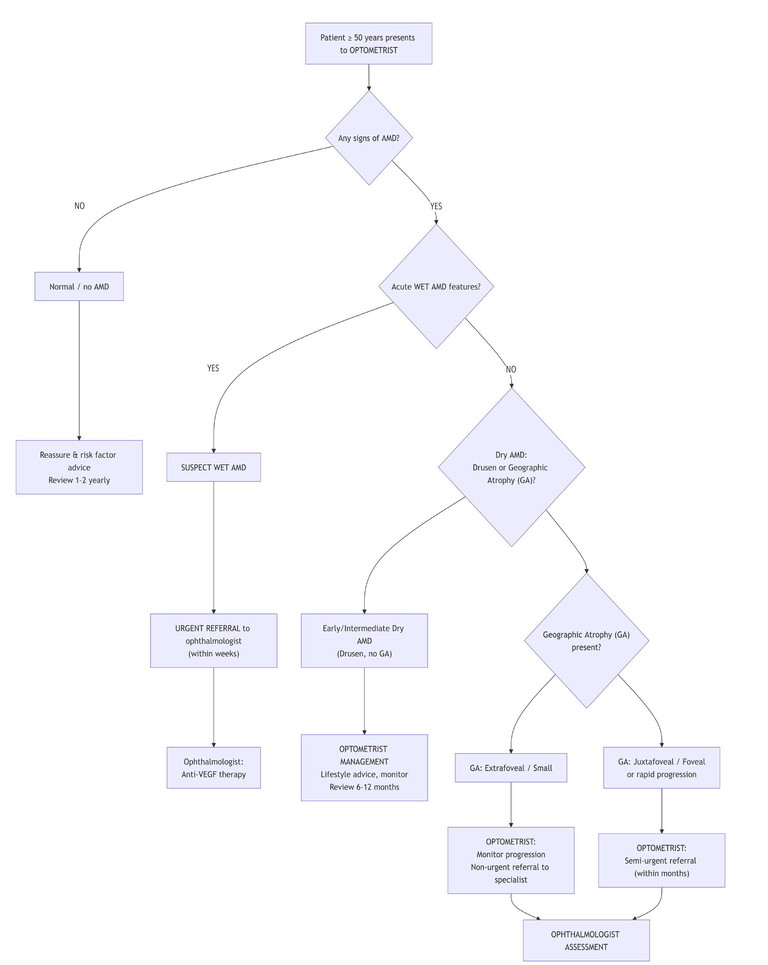
Table 2: An example of a referral pathway for patients with AMD.
Administration (TGA) in Australia for GA secondary to AMD.9-13 These agents do not restore lost tissue or, on average, improve visual acuity, but they consistently slow the rate of GA lesion enlargement compared with sham injections in Phase 3 trials.9-12,14
One way to explain this to patients: “None of today’s treatments can ‘re-grow’ the damaged cells, but they can slow the disease marching further into your central vision”, or, if using the lawn analogy, “they can stop a bare patch of grass spreading to destroy the whole lawn – buying more good reading and driving years than you would otherwise have”.14,15
IDEAL CANDIDATES AND MECHANISM OF COMPLEMENT INHIBITORS
Q. In an ideal world, who would be the best candidate for complement inhibitors to slow the progression of GA, and what is the mechanism of action?
Complement inhibitors target an overactive component of innate immunity, the complement cascade, which plays a major role in chronic inflammation and cell death in GA.7,16 Pegcetacoplan binds C3, a central ‘hub’ of the cascade, thereby reducing downstream activation, while avacincaptad pegol binds C5, aiming to reduce the inflammatory and cell-killing components (including membrane attack complex formation) that drive atrophy while preserving some upstream complement function.9,11,12,16
In OAKS and DERBY, monthly or every-other-month pegcetacoplan reduced GA growth by roughly 16–21% at 12 months and about 19–22% at 24 months versus sham, with greater effects in non-subfoveal lesions and over longer follow up.9,10 In GATHER1 and GATHER2, monthly avacincaptad pegol reduced GA expansion by around 18–35% at 12 months, depending on lesion location and analysis, with benefits sustained over two years.11,12 Extension and emerging real-world data suggest that benefit accrues over time, at the cost of ongoing regular intravitreal injections.10,14,17 It is important to remember that there is currently no head-to-head trial between the two molecules. Additionally, the main trials of the two molecules have different inclusion and exclusion criteria, hence their data cannot be compared directly, but for the convenience of an overview, please see Table 1.
Q. So, who is the ‘ideal candidate?
The archetypal candidate is an older adult with GA secondary to AMD, sufficiently preserved visual acuity to gain from preserving function, and lesions that threaten, but have not yet destroyed the fovea – especially extrafoveal or juxtafoveal GA where preserving central vision is most realistic.7,14,18 They must be motivated, able to attend monthly or every-other-month injections long-term, and willing to accept a higher risk of conversion to neovascular AMD and injection-related complications compared with observation alone.9,14,18 Most crucially, they need to comprehend that these treatments will not improve their vision, unlike the anti-VEGF treatments for wet AMD.
A metaphor for the mechanism: “Think of complement as an over-sensitive security system. In GA, the alarm keeps going off, and the sprinklers soak healthy rooms along with the damaged ones. These drugs dial the system down, so it still works but doesn’t flood the whole floor every time.”7,18
WHEN TO REFER AND WHAT TO INCLUDE
Q. When should optometrists refer patients for consideration of complement inhibitor treatment, and what three important details should be included in the referral?
Updated RANZCO (Royal Australian and New Zealand College of Ophthalmologists) and Optometry Australia pathways encourage optometrists to identify patients with GA or nGA and discuss potential treatment options as therapies become available.1,2,8 Referral is generally appropriate when you detect definite GA lesions (particularly extrafoveal or juxtafoveal) in a patient with meaningful functional vision, or when GA is progressing towards the fovea on serial imaging.2,7,8,18
Three core elements to include are: Clear clinical description and staging. Best-corrected visual acuity, AMD stage in each eye, GA location and approximate size (e.g. disc diameter or square millimetres if available), and symptoms such as scotomas or reading difficulty.1,8
Imaging. OCT (and FAF or colour photos if available) showing drusen, RPE changes, GA boundaries, and any signs of neovascular conversion (subretinal/intraretinal fluid, haemorrhage).1-5
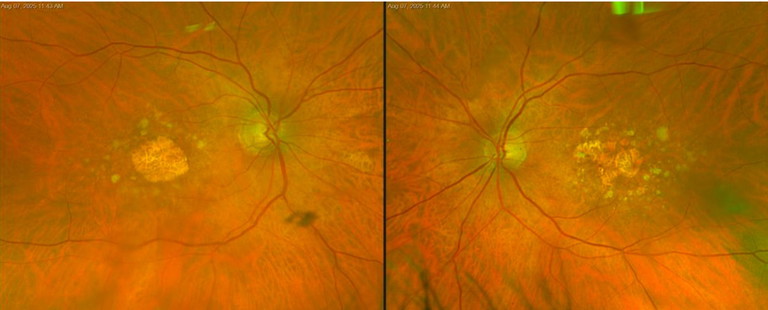
Figure 1. Colour fundus photographs showing area with GA with surrounding drusen.
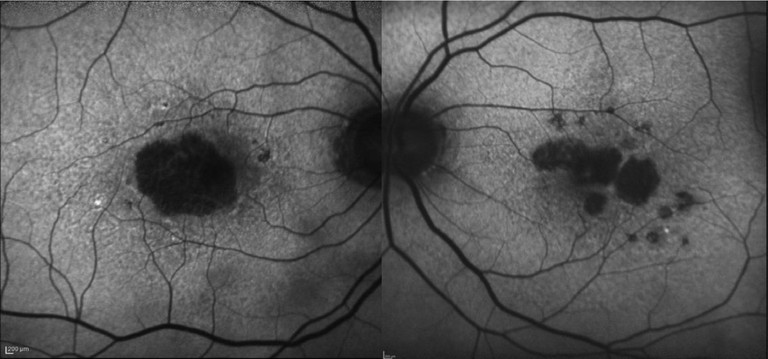
Figure 2. Fundus autofluorescence images showing area with GA.
Risk factors and systemic context. Smoking status, cardiovascular risk factors, prior intravitreal therapy, anticoagulation, mobility/ transport issues, and patient goals (driving, reading, caring responsibilities).2,8,16,17
These details allow the retinal specialist to judge eligibility, then balance risks and benefits, and plan a realistic injection schedule tailored to the patient’s circumstances.15,17
EXPLAINING BENEFITS IN PATIENT-FRIENDLY TERMS
Q. How do you explain the benefits of complement inhibitors to patients so that they understand the importance of treatment?
Many patients expect a ‘fix’, so the first step is to reframe the goal from ‘improving vision’ to ‘preserving vision for longer’.15,16 A helpful analogy compares GA to a bushfire heading towards a town – we cannot rebuild houses already burned, but we can build firebreaks and slow the fire so it reaches fewer homes and arrives much later, giving more years of safe living.15
In large clinical trials, patients receiving complement inhibitors lost less healthy retinal tissue over time than sham-treated patients, translating into more years of useful reading, face recognition, and independent function.9-12,14 It is important to explain that benefit accumulates gradually – there is usually no day-to-day improvement to ‘feel’ – but long-term, in the order of years, the treated eye tends to do better than if left untreated.10,14,17
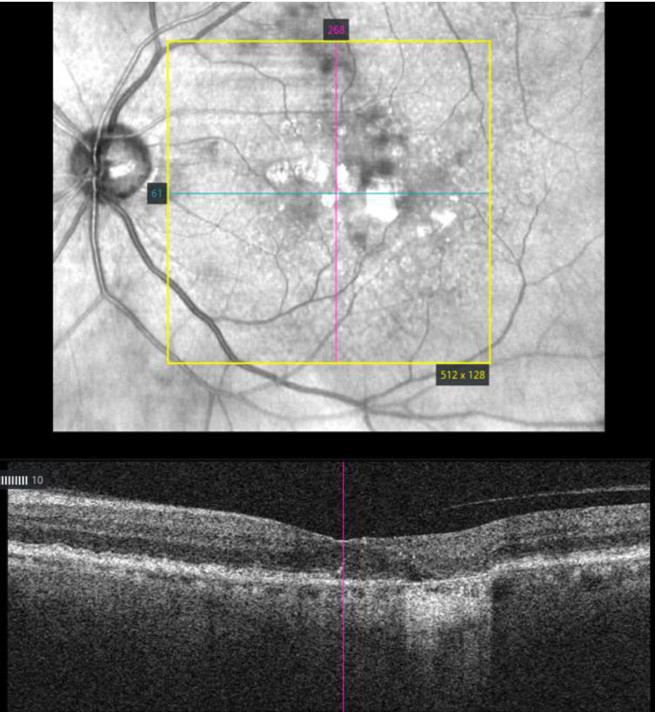
Figure 3. HD-OCT raster scan showing an area of nGA and GA.
A description you can adapt: “Your scans show an area of ‘wear and tear’ in the centre of your retina that will slowly enlarge over time; this injection can’t patch the worn area, but it can slow how fast it spreads; we aim to keep you reading, recognising faces, and managing daily tasks for as long as we can.”15,16
MANAGING EXPECTATIONS WHEN VISUAL ACUITY DOESN’T IMPROVE
Q. Given that the complement inhibitors slow GA lesion growth but do not improve visual acuity in the clinic studies, how do you manage patient expectations?
Expectation management is crucial; most trial participants did not show an average improvement in visual acuity, so patients should be told clearly that letter scores on the chart are unlikely to go up with treatment.9-12,14 The value lies in losing vision more slowly – shifting the conversation from “Will I see better?” to “Will I see for longer?”.
One useful comparison for patients is between GA treatment and cholesterol tablets: “You do not feel better after each pill, but over the years, you are less likely to have a major event.”15,18 Functional assessments, such as microperimetry, suggest better preservation of retinal sensitivity near the GA border in treated eyes, reinforcing that treatment is protecting function, even when acuity is static.10,14,18
“... most GA is first detected in optometry, often before symptoms are prominent”
A short phrase to use: “This treatment is about slowing the clock, not turning it back. At least, you potentially enjoy a longer period of driving ability, maintaining independence, enjoying your hobbies, and lastly, more time to prepare for changes in your vision.”15,16
EXPLAINING THE LONG-TERM TREATMENT COMMITMENT
Q. What is your advice to optometrists when explaining the long-term treatment commitment associated with complement inhibitor therapy?
Patients need to understand from the outset that current regimens involve injections into the eye every month or every second month, and that most benefit appears to accrue with continuous, long-term treatment rather than short ‘bursts’.9-12,14 Clinical and qualitative data suggest that persistence is challenging; acceptability falls when injection frequency, travel burden, and perceived benefit feel out of balance.16,17
It can be useful to liken the regimen to regular dialysis or insulin – an ongoing treatment that keeps a chronic disease from progressing as fast as it otherwise would.12,18 Encourage practical planning: discuss who can drive them, which time of day works best, and what contingency plans exist if a caregiver is unavailable, so adherence is woven into the routine rather than relying purely on motivation.16,17
A phrasing you can use: “To get the most out of this treatment, we need to keep topping up the medication regularly – the same way that you keep topping up fuel in a car on a long trip. Missing the odd injection happens, but stopping altogether usually means the disease goes back to its natural faster speed.15,16
DISCUSSING COST, INSURANCE AND ACCESS
Q. What should optometrists tell patients about treatment cost, insurance coverage, and access pathways?
These drugs are high-cost biologics. Coverage arrangements vary between countries and between public and private sectors, and will evolve as payers review cost-effectiveness.13,18 In Australia, pegcetacoplan and avacincaptad pegol are now registered for GA secondary to AMD, but funding and out-of-pocket costs differ between public hospitals, private retina clinics, and insurers, so it is safest to advise that the retinal clinic will provide up-to-date cost information and subsidy options.12,13 Macular Disease Foundation Australia provides valuable resources in answering these questions that patients may have. Dr Kathy Chapman, the CEO, and her team are great patient advocates.
Payer analyses note that decision makers weigh the modest relative reduction in GA progression against treatment cost, injection frequency, and safety profile, which may translate into prior authorisation or specific eligibility criteria.13,18 From the patient’s perspective, qualitative studies indicate that perceived functional benefit, risk of side effects, travel burden, and cost, all influence willingness to start and continue complement inhibitor therapy.17
A chairside talking point: “Because these are new, specialised medicines, the exact cost to you depends on which clinic you attend and your insurance or pension status. The retina clinic will walk you through the fees and any subsidy options before you decide – our role is to flag this option and make sure the right team sees you.”12,13
OPTOMETRISTS’ RESPONSIBILITIES IN SHARED CARE
Q. What are optometrists’ key responsibilities, within a shared-care model, to ensure effective management of patients with GA?
RANZCO and Optometry Australia documents place community optometrists at the front line of GA care: most GA is first detected in optometry, often before symptoms are prominent.2,8 Key responsibilities include accurate AMD staging, documenting, and monitoring GA over time with OCT and, where possible, FAF, and identifying high-risk features such as reticular pseudodrusen or nascent GA.1-5
Once a patient is co-managed with a retinal specialist, optometrists play a vital role in reinforcing education, checking adherence and satisfaction, monitoring for conversion to neovascular AMD (new fluid, haemorrhage or sudden distortion), and promptly communicating any changes.2,8,16 Be aware that patients on the new complement inhibitors have a higher incidence of new choroidal neovascular membrane compared to patients on sham injections, but they can have anti-VEGF injections as well, as in the clinical trials (Table 2).9,12
Optometrists also coordinate low-vision and rehabilitation referrals, maintain general ocular care (refractive updates, cataract co-management, dry eye), and liaise with the patient’s general practitioner about systemic risk-factor control.1,2,6,8
A way to frame this to colleagues and patients: “Think of GA care as a relay: the optometrist spots the problem early and hands the patient to the retinal specialist for treatment, but then keeps running alongside – checking the map, watching for new warning signs, and helping ensure the patient does not drop out of the race.”1,2,8
This interview was conducted at the Queensland Eye Institute, Brisbane.
The sponsor of this article is Astellas. Astellas Australia had no direct input on the answers to the questions.
To earn your CPD hours from this article, scan the QR code or visit: mieducation.com/managing-dry-age-related-macular-degeneration-a-fireside chat-with-associate professoranthony-kwan.

References available at mieducation.com.

Associate Professor Anthony Kwan MBChB (UK) MD (London) FRCOphth (UK) FRANZCO is a vitreoretinal surgeon, retinal specialist, and the Director of Vitreoretinal Service at the Queensland Eye Institute. He is a senior staff specialist and the former Head of Ophthalmology at Mater Hospital, Brisbane.
Assoc Prof Kwan teaches medical students and ophthalmic trainees at the Mater Hospital, and is a training post inspector for the Royal Australian and New Zealand College of Ophthalmologists (RANZCO). He is on the advisory board of numerous pharmaceutical companies and is an active organiser for national and international ophthalmic meetings.

Tina Huynh BAppSci (Optom) (Hons 1st) GradCertOcThera is the Clinical Operations Coordinator at Queensland Eye Institute and a practising optometrist.